Targeting Pathogens: Advances in Bacteria-Specific Molecular Imaging Probes for Precision Infection Diagnosis
This article provides a comprehensive review of the rapidly evolving field of bacteria-specific molecular imaging probes for infection localization.
Targeting Pathogens: Advances in Bacteria-Specific Molecular Imaging Probes for Precision Infection Diagnosis
Abstract
This article provides a comprehensive review of the rapidly evolving field of bacteria-specific molecular imaging probes for infection localization. We first establish the critical clinical need for distinguishing bacterial infections from sterile inflammation and explore the foundational principles of probe design, including target selection and probe chemistry. We then detail current methodological approaches, from antibiotic-derived agents to metabolic probes and smart activatable systems, highlighting their translational applications. A dedicated section addresses common challenges in probe development, such as improving specificity, pharmacokinetics, and signal-to-noise ratios. Finally, we compare and validate leading probe strategies against clinical standards like PET/CT with FDG and white blood cell scintigraphy. This synthesis is intended for researchers, scientists, and drug development professionals seeking to understand the state-of-the-art and future trajectory of this transformative diagnostic technology.
The Why and How: Foundational Principles of Bacterial Targeting for Imaging
The clinical challenge of distinguishing bacterial infection from sterile inflammation is a critical diagnostic imperative. Misdiagnosis leads to antimicrobial misuse, increased resistance, and poor patient outcomes. This application note, framed within a broader thesis on bacteria-specific molecular imaging probes, details current methodologies, quantitative biomarkers, and experimental protocols to address this challenge. The focus is on research tools that enable precise localization and identification of live pathogens in vivo.
Quantitative Biomarkers: Infection vs. Sterile Inflammation
Current research identifies key molecular and cellular markers with differential expression. The following tables summarize quantitative data from recent studies (2023-2024).
Table 1: Serum Protein Biomarkers
| Biomarker | Typical Range in Sterile Inflammation | Typical Range in Bacterial Infection | AUC (95% CI) from Recent Meta-Analysis | Key Differentiating Factor |
|---|---|---|---|---|
| Procalcitonin (PCT) | <0.5 ng/mL | >0.5 ng/mL (can be >>2 ng/mL) | 0.85 (0.82–0.88) | Sharp rise with bacterial sepsis; minimal in viral/sterile. |
| C-Reactive Protein (CRP) | Elevated (10-100 mg/L) | Highly Elevated (often >100 mg/L) | 0.72 (0.68–0.76) | Poor specificity alone; kinetics useful. |
| Lipocalin-2 (NGAL) | Moderately Elevated | Highly Elevated in Gram-negative | 0.79 (0.74–0.83) | Binds bacterial siderophores. |
| Presepsin (sCD14-ST) | <500 pg/mL | >500 pg/mL | 0.88 (0.85–0.91) | Specific to phagocyte response to pathogens. |
| IL-6 | Very High (often >500 pg/mL) | High (100-1000 pg/mL) | 0.65 (0.60–0.70) | Higher in cytokine storms (sterile). |
Table 2: Imaging Probe Performance Metrics
| Probe Type/ Target | Model System | Signal-to-Background Ratio (Infection) | Signal-to-Background Ratio (Sterile Inflammation) | Specificity (vs Sterile) | Reference Year |
|---|---|---|---|---|---|
| [18F]FDG-PET | Mouse, myositis | 3.2 ± 0.5 | 2.8 ± 0.6 | Low | 2023 |
| [68Ga]Ga-citrate-PET | Rat, abscess | 5.1 ± 1.2 | 1.9 ± 0.3 | High | 2023 |
| Maltodextrin-based fluorescent probe (Mal-Cy5) | Mouse, S. aureus | 8.5 ± 2.1 | 1.5 ± 0.4 | Very High | 2024 |
| Vancomycin-IRDye800CW | Mouse, implant | 6.7 ± 1.8 | 2.2 ± 0.5 | High | 2023 |
| Specific probe for Bacterial protease (PGF-1) | Mouse, E. coli | 9.2 ± 1.5 | 1.3 ± 0.2 | Very High | 2024 |
Experimental Protocols
Protocol 3.1:In VivoValidation of a Bacteria-Specific Maltodextrin Probe
Objective: To differentiate Staphylococcus aureus infection from sterile inflammation (e.g., zymosan-induced) in a murine model using fluorescence imaging.
Materials: See "The Scientist's Toolkit" (Section 5). Procedure:
- Animal Model Preparation:
- Infection Group (n=5): Anesthetize mice. Inject 50 µL of PBS containing 1x10^7 CFU of log-phase S. aureus intramuscularly into the right hind limb.
- Sterile Inflammation Group (n=5): Inject 50 µL of PBS containing 100 µg of zymosan A into the left hind limb.
- Control Group (n=3): Inject 50 µL of sterile PBS.
- Probe Administration: At 24h post-induction, inject 2 nmol of the maltodextrin-based fluorescent probe (e.g., Mal-Cy5) via the tail vein.
- In Vivo Imaging:
- At 2h and 24h post-injection, anesthetize mice.
- Acquire fluorescence images using a preclinical imager (e.g., PerkinElmer IVIS or Li-COR Pearl).
- Use consistent settings: excitation 640 nm, emission 700 nm, medium binning, 5-second exposure.
- Shave imaging area to reduce autofluorescence.
- Ex Vivo Analysis:
- Euthanize mice after final imaging. Harvest tissues (muscle at injection site, liver, spleen, kidney).
- Image tissues ex vivo using the same settings.
- Quantify mean fluorescence intensity (MFI) within a standardized region of interest (ROI).
- Homogenize the infected/inflamed tissue and perform serial dilution plating on agar to determine bacterial CFU.
- Data Analysis:
- Calculate target-to-background ratio (TBR) as: TBR = MFI (target tissue) / MFI (contralateral muscle or control tissue).
- Correlate TBR with CFU counts. Perform statistical analysis (unpaired t-test) between infection and sterile inflammation groups.
Protocol 3.2: Multiplexed Cytokine/Presepsin Assay for Serum Profiling
Objective: To quantify a panel of serum biomarkers (Presepsin, IL-6, IL-10, TNF-α) to create a diagnostic signature.
Materials: Multiplex bead-based immunoassay kit (e.g., Luminex), serum samples, plate reader. Procedure:
- Sample Collection: Collect blood via terminal cardiac puncture or submandibular bleed. Allow clotting (30 min, RT). Centrifuge at 2000 x g for 10 min. Aliquot serum and store at -80°C.
- Assay Setup:
- Thaw samples on ice. Prepare standards, controls, and wash buffer per kit instructions.
- Add 50 µL of standards, controls, and diluted serum (1:4) to a 96-well filter plate containing capture antibody-coupled beads.
- Seal and incubate on a plate shaker (850 rpm) for 2h at RT, protected from light.
- Detection:
- Aspirate liquid. Wash wells 3x with wash buffer.
- Add 50 µL of biotinylated detection antibody mixture. Incubate for 1h with shaking.
- Wash 3x. Add 50 µL of streptavidin-PE. Incubate for 30 min with shaking.
- Wash 3x. Resuspend beads in 100 µL of reading buffer.
- Acquisition & Analysis:
- Read plate on a Luminex analyzer within 90 minutes.
- Use assay software to generate a 5-parameter logistic standard curve for each analyte.
- Interpolate sample concentrations. Apply dilution factor.
Visualizing Pathways and Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Infection Imaging Research
| Item Name | Supplier Examples (Catalog #) | Function in Research | Key Consideration |
|---|---|---|---|
| Maltodextrin-Based Imaging Probe (Mal-Cy5) | Custom synthesis (e.g., AAT Bioquest) | Fluorescent probe selectively transported into bacteria via maltodextrin transporter. High specificity over mammalian cells. | Requires validation for bacterial strain of interest. Optimal imaging window ~2-6h post-injection. |
| Vancomycin-Fluorophore Conjugate | PerkinElmer (NEV100XX) | Binds to D-Ala-D-Ala peptidoglycan precursors in Gram-positive bacteria. Useful for surgical guidance. | Binds both live and dead bacteria. Potential renal uptake. |
| PGF-1 Fluorescent Substrate | Cayman Chemical (25835) | Activated by bacterial protease (e.g., E. coli Plasmepsin IV). Offers enzymatic amplification. | Specificity profile must be matched to infecting pathogen. |
| [68Ga]Ga-Citrate | Radio-pharmacy (GMP grade) | PET tracer that acts as an iron analog, taken up by bacteria via siderophore systems and by host lactoferrin. | Requires on-site cyclotron/generator. Excellent for deep-tissue infection. |
| Luminex Multiplex Assay Kit (Human Sepsis Panel) | R&D Systems (LXSAHM) | Simultaneously quantifies 10+ serum biomarkers (Presepsin, PCT, cytokines) from small sample volume. | Enables signature-based diagnosis. Requires Luminex platform. |
| Zymosan A, from S. cerevisiae | Sigma-Aldrich (Z4250) | Induces potent sterile inflammation via TLR2/dectin-1. Standard for control sterile inflammation models. | Prepare fresh suspensions. Dose varies by route (10-100 µg i.m.). |
| IVIS Spectrum In Vivo Imager | PerkinElmer | Enables 2D/3D fluorescence and bioluminescence imaging. Critical for probe kinetics and biodistribution. | Choose appropriate filter sets for probe. Maintain consistent anesthesia. |
| CFW (Calcofluor White) | Sigma-Aldrich (F3543) | Fluorescent dye binding to chitin and cellulose. Stains fungal cell walls; control for fungal infection models. | Not specific to bacteria. Useful as a counterstain or fungal model agent. |
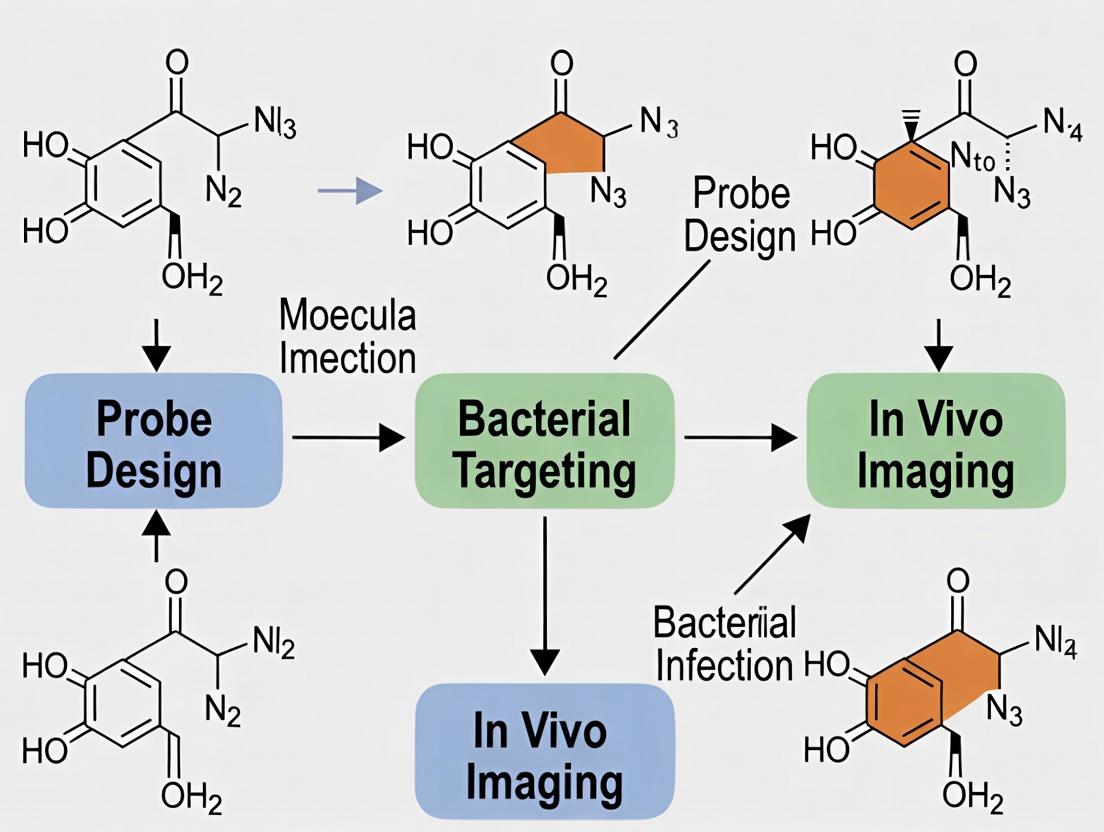
Within the critical research field of bacteria-specific molecular imaging for infection localization, the design of an effective probe is foundational. This application note details the core structural components and guiding design principles essential for constructing probes that can selectively distinguish bacterial infections from sterile inflammation, thereby advancing diagnostic and therapeutic development.
Core Components of a Bacteria-Specific Imaging Probe
An effective probe is a modular assembly of distinct functional units. The integration of these components determines specificity, signal fidelity, and pharmacokinetic profile.
Targeting Vector/Moiety
This component confers specificity by binding to a unique molecular signature of the target pathogen.
- Common Targets: Bacterial surface enzymes (e.g., sortase A), metabolic pathways (e.g., D-amino acids, maltodextrin), upregulated host responses (e.g., antimicrobial peptides).
- Types: Small molecules, peptides, peptidomimetics, antibody fragments (e.g., single-domain VHHs).
Signaling Unit/Reporter
This moiety generates the detectable signal upon successful probe localization.
- Optical Imaging: Fluorophores (e.g., Cy5, ICG, NIR-II dyes).
- Nuclear Imaging: Radioisotopes for PET (e.g., ⁶⁸Ga, ¹⁸F) or SPECT (e.g., ⁹⁹ᵐTc).
- Other: MRI contrast agents (e.g., Gd³⁺ chelates), acoustic contrast agents.
Linker/Spacer
A chemical bridge connecting the targeting and signaling units. It is critical for maintaining the binding affinity of the vector and the functionality of the reporter. Linkers can be cleavable (enzymatically or by pH) or non-cleavable.
Pharmacokinetic Modifier
Optional elements that adjust the probe's in vivo behavior, such as polyethylene glycol (PEG) chains to enhance circulation half-life or reduce non-specific uptake.
Key Design Principles for Infection Imaging
The following principles guide probe development for high-contrast bacterial localization.
Table 1: Core Design Principles for Bacterial Imaging Probes
| Principle | Objective | Key Considerations |
|---|---|---|
| High Specificity & Affinity | Maximize target-to-background ratio by discriminating bacterial from host cells. | Target selection (bacteria-specific vs. host-response); Binding affinity (Kd in nM range). |
| Signal Activation/Amplification | Generate signal primarily at the site of infection to improve sensitivity. | Use of activatable ("smart") probes quenched until cleaved by bacterial enzymes. |
| Favorable Pharmacokinetics | Rapid clearance from non-target tissues with retention at the infection site. | Molecular size, charge, hydrophilicity; Renal vs. hepatic clearance pathways. |
| Minimal Immunogenicity & Toxicity | Ensure biocompatibility for potential clinical translation. | Use of humanized or small targeting ligands; stable, non-toxic linkers and reporters. |
Experimental Protocol: Evaluating a Fluorophore-Quencher Based Activatable Probe
This protocol details the in vitro validation of a peptide-based NIRF probe activated by bacterial protease cleavage.
Protocol 1: In Vitro Activation Assay
Objective: To confirm specific activation of the probe by target bacterial enzyme versus mammalian proteases.
Materials:
- Synthesized Probe: Target peptide sequence, conjugated to NIR fluorophore (e.g., Cy5.5) and a quencher (e.g., QSY21) via a cleavable linker.
- Enzymes: Recombinant target bacterial enzyme (e.g., Sortase A), control mammalian protease (e.g., MMP-9), reaction buffers.
- Equipment: Fluorescence microplate reader, incubation shaker, HPLC system for fragment analysis.
Procedure:
- Sample Preparation: Dilute the probe to 1 µM in appropriate assay buffer (pH 7.4). Prepare 10 µL of each enzyme solution at 100 nM.
- Reaction Setup: In a black 96-well plate, mix 90 µL of probe solution with 10 µL of: a) Target bacterial enzyme, b) Control mammalian enzyme, c) Buffer only (negative control). Perform in triplicate.
- Incubation: Incubate plate at 37°C with gentle shaking.
- Kinetic Readout: Measure fluorescence intensity (Ex/Em per Cy5.5) every 5 minutes for 2 hours using a plate reader.
- Terminal Analysis: After 2 hours, analyze 20 µL from each reaction by analytical HPLC to separate and confirm the generation of the fluorescent fragment.
Data Analysis: Plot fluorescence intensity over time. Calculate the fold-increase in signal for the bacterial enzyme sample relative to controls. HPLC chromatograms should show a peak corresponding to the cleaved fluorophore-labeled fragment only in the active enzyme sample.
Table 2: Representative Kinetic Data from Activation Assay
| Time (min) | Fluorescence Intensity (RFU, Mean ± SD) | ||
|---|---|---|---|
| Probe + Target Enzyme | Probe + Control Enzyme | Probe Only | |
| 0 | 550 ± 45 | 520 ± 38 | 510 ± 42 |
| 30 | 4,850 ± 210 | 810 ± 65 | 580 ± 55 |
| 60 | 12,300 ± 540 | 950 ± 72 | 605 ± 58 |
| 120 | 18,950 ± 880 | 1,100 ± 98 | 620 ± 60 |
Diagram: Probe Activation and Imaging Workflow
Title: Mechanism of Bacteria-Activated Probe Imaging
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Research Reagents for Probe Development & Validation
| Reagent/Category | Function & Rationale |
|---|---|
| D-Amino Acid-Based Probes (e.g., FDAAs) | Incorporate into bacterial cell wall peptidoglycan via transpeptidases, providing a highly specific labeling strategy for live bacteria. |
| Sortase A Substrate Peptides | Serve as targeting vectors for Gram-positive bacteria; cleaved and covalently incorporated by the Sortase A enzyme. |
| NIR-II Fluorophores (e.g., CH-4T) | Enable deeper tissue penetration and higher resolution in vivo optical imaging due to reduced scattering in the second near-infrared window. |
| Chelators for Radiometals (e.g., DOTA, NOTA) | Bind diagnostic radioisotopes (⁶⁸Ga, ⁶⁴Cu) for PET imaging, forming a stable complex in vivo for accurate infection tracing. |
| Quencher Dyes (e.g., QSY21, BBQ-650) | Suppress fluorophore emission via FRET when in close proximity, used in constructing "off-on" activatable probes for low-background imaging. |
| Mycobacterium-Specific Siderophores | Iron-chelating compounds repurposed as targeting moieties for imaging tuberculosis and other mycobacterial infections. |
| Antimicrobial Peptide Derivatives (e.g., UBI) | Bind to negatively charged bacterial membranes; fragments can be engineered for selective uptake in infected versus mammalian cells. |
Application Notes & Protocols for Infection Localization Research
This document presents key protocols and application notes for developing molecular imaging probes targeting bacteria-specific biomarkers. Framed within a thesis on infection localization, the focus is on three target classes: the bacterial cell wall (peptidoglycan, mycolic acid), core metabolic pathways (folate synthesis, siderophore systems), and enzymes (β-lactamases, sortases). These non-mammalian targets enable specific in vivo detection of bacterial infections, distinguishing them from sterile inflammation—a critical challenge in diagnostic imaging and therapeutic monitoring.
Table 1: Key Bacterial-Specific Biomarkers and Probe Development Status
| Biomarker Class | Specific Target | Pathogen Examples | Known Targeting Ligand/Probe (Example) | Reported In Vivo Imaging Modality | Key Challenge (Selectivity/Sensitivity) |
|---|---|---|---|---|---|
| Cell Wall | Peptidoglycan (Strain-specific sugars) | S. aureus, E. coli | Fluorescent D-amino acids (FDAAs), Vancomycin-fluorophore conjugates | Optical (NIRF), PET ([18F]FDG-analogs) | Permeability in Gram-negatives; mammalian cell background. |
| Cell Wall | Mycolic Acid | M. tuberculosis | [11C]Para-aminobenzoic acid (PABA), Trehalose analogs | PET, SPECT | Slow bacterial growth rate limits signal accumulation. |
| Metabolism | Dihydrofolate Reductase (DHFR) | Trimethoprim-sensitive species | Trimethoprim-based probes (TMP-[Near-IR dye]) | Optical (NIRF) | Human DHFR off-target binding must be engineered out. |
| Metabolism | Siderophore Receptors (e.g., FhuA) | P. aeruginosa, K. pneumoniae | Ferrioxamine-68Ga, Pyochelin-99mTc | PET, SPECT | Complexity of siderophore synthesis and conjugation. |
| Enzymes | β-Lactamase (e.g., TEM-1) | Resistant Enterobacteriaceae | Cefalosporin-based activatable probes (e.g., CCF2/4-AM) | Optical (FRET), PET | Requires enzyme presence; not constitutive in all strains. |
| Enzymes | Sortase A (SrtA) | S. aureus | LPETG peptide sequence with quenched fluorophore | Optical (NIRF) | Extracellular activity; potential cleavage by host proteases. |
Table 2: Recent In Vivo Performance Metrics for Selected Probes
| Probe Name | Target | Model (Mouse) | Pathogen | Signal-to-Background Ratio (T/NT) | Time to Peak Signal | Reference Year* |
|---|---|---|---|---|---|---|
| Fluor-D-Lys( Cy5) | Peptidoglycan synthesis | Thigh infection | S. aureus | 3.8 ± 0.4 | 6 h | 2023 |
| 68Ga-FSC | Siderophore receptor | Lung infection | P. aeruginosa | 5.2 ± 1.1 | 2 h | 2024 |
| TMP-IR800 | Bacterial DHFR | Myositis | E. coli | 4.1 ± 0.7 | 24 h | 2022 |
| Activatable β-Lactamase Probe (NIR) | β-Lactamase | Subcutaneous abscess | E. coli (TEM-1+) | 8.5 ± 1.2 (Activ. Ratio) | 90 min | 2023 |
Note: Data synthesized from recent literature searches.
Detailed Experimental Protocols
Protocol 1: Synthesis and Purification of a Siderophore-68Ga Conjugate for PET Imaging
Objective: To radiolabel the hydroxamate siderophore deferoxamine (DFO) with Gallium-68 for in vivo PET imaging of siderophore receptor-positive bacteria.
I. Materials (Research Reagent Solutions)
- DFO-p-SCN (Macrocyclics, #B-705): Chelator precursor for conjugation.
- Siderophore Analogue (e.g., Fusarinine C, custom synthesis): Bacterial iron chelator for specific targeting.
- 0.1 M Sodium Acetate Buffer (pH 4.5): Reaction buffer for optimal 68Ga labeling.
- 68Ga/68Ge Generator (e.g., ITG GmbH): Source of positron-emitting 68Ga.
- PD-10 Desalting Column (Cytiva): For rapid purification of the radiolabeled conjugate.
- Radio-HPLC System: Equipped with a UV/Vis and radioactivity detector for quality control.
- Sterile 0.9% NaCl, 0.1% Tween 80 Solution: For final formulation.
II. Procedure
- Conjugation: Dissolve 1 mg of the siderophore analogue (amine-functionalized) in 200 µL of 0.1 M carbonate buffer (pH 9.0). Add a 1.5 molar excess of DFO-p-SCN dissolved in DMSO. React for 2 h at room temperature with gentle shaking.
- Purification of Conjugate: Purify the DFO-siderophore conjugate using semi-preparative HPLC. Lyophilize the pure fraction and store at -20°C.
- 68Ga Radiolabeling: a. Elute 68Ga3+ from the generator with 0.1 M HCl into a reaction vial. b. Adjust the eluate to pH ~4.0 using 1 M sodium acetate buffer (pH 4.5). c. Add 10-50 µg of the DFO-siderophore conjugate. d. Heat at 95°C for 10 minutes.
- Purification & Formulation: Pass the reaction mixture through a pre-conditioned PD-10 column, eluting with sterile saline. Collect the radioactive fraction containing the labeled conjugate. Pass through a 0.22 µm sterile filter into a final vial. Perform quality control using Radio-TLC (ITLC) and Radio-HPLC. Radiochemical purity should be >95%.
Protocol 2: Ex Vivo Validation of Probe Specificity Using Infected Tissue Homogenates
Objective: To confirm enzymatic activation of a β-lactamase-sensitive probe in homogenates from infected vs. inflamed tissue.
I. Materials
- Activatable Probe (e.g., NIR-cephalosporin-quencher conjugate): Synthesized in-house or commercially sourced.
- Infected Tissue Model: Thigh muscles from mice infected with β-lactamase-positive bacteria (e.g., TEM-1 E. coli).
- Control Tissues: From mice with: a) sterile inflammation (e.g., LPS), b) β-lactamase-negative infection.
- Homogenization Buffer: PBS with 1% Triton X-100 and protease inhibitor cocktail.
- Microplate Reader/ Fluorescence Imager: Capable of detecting the probe's emission wavelength (e.g., 700 nm for NIR).
II. Procedure
- Tissue Homogenate Preparation: Sacrifice animals at peak infection/inflammation (e.g., 24 h post inoculation). Excise and weigh target tissues. Homogenize in ice-cold buffer (100 mg tissue/mL) using a bead-beater or mechanical homogenizer. Clarify by centrifugation (10,000 x g, 10 min, 4°C). Collect supernatant.
- Fluorescence Activation Assay: a. In a black 96-well plate, add 90 µL of each tissue homogenate supernatant (in triplicate). b. Add 10 µL of the activatable probe solution (final concentration 1 µM). c. Include control wells: Probe + Lysis Buffer (background), Probe + Purified TEM-1 Enzyme (positive control). d. Incubate plate at 37°C for 60 minutes. e. Measure fluorescence (Ex/Em per probe specifications, e.g., 680/720 nm) at time zero and every 15 minutes.
- Data Analysis: Calculate fluorescence fold-increase relative to time zero and background. Compare final signals between infected (target enzyme-positive), inflamed, and negative infection groups. Statistical significance is typically assessed via one-way ANOVA.
Visualizations: Pathways & Workflows
Diagram 1: Bacterial Cell Wall Synthesis & Probe Incorporation Pathways
Diagram 2: Workflow for Developing an Enzymatically-Activated Imaging Probe
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Bacterial Biomarker Probe Development
| Reagent / Material | Supplier Examples | Primary Function in Research |
|---|---|---|
| Fluorescent D-Amino Acids (FDAAs, HADA, NADA) | Custom synthesis (e.g., Sigma-Aldrich custom service) | Direct incorporation into bacterial peptidoglycan for labeling and imaging cell wall synthesis. |
| DFO-p-SCN / NOTA-p-SCN / DOTA-NHS | Macrocyclics, CheMatech | Bifunctional chelators for conjugating targeting vectors to radiometals (68Ga, 64Cu, 111In). |
| Siderophore Analogs (e.g., Enterobactin, Pyoverdine cores) | EMC Microcollections, custom synthesis | High-affinity targeting ligands for bacterial iron-acquisition systems. |
| β-Lactamase Substrate Scaffolds (Cephalosporin core) | Tocris, Fisher Scientific, custom | Backbone for designing enzyme-activated (smart) probes that cleave upon enzyme exposure. |
| Near-Infrared Fluorophores (e.g., IRDye 800CW, Cy7) | LI-COR, Lumiprobe, Cyandye | Reporter dyes for optical imaging, offering deep tissue penetration and low autofluorescence. |
| Sortase A Substrate Peptides (e.g., LPETGG-amide) | Genscript, Peptide 2.0 | Peptide sequences for probing or exploiting bacterial surface protein anchoring activity. |
| Mycolic Acid / Trehalose Analogs | Carbosynth, Avanti Polar Lipids | Precursors for probing unique mycobacterial cell wall components. |
| Trimethoprim (TMP) Analogs with Reactive Handles | Sigma-Aldrich, modified in-house | Scaffold for targeting bacterial dihydrofolate reductase (DHFR). |
Within the thesis on bacteria-specific molecular imaging probes for infection localization, selecting the appropriate imaging modality is paramount. Each modality offers distinct advantages in sensitivity, resolution, quantification, and clinical translation. This document provides application notes and detailed experimental protocols for utilizing Positron Emission Tomography (PET), Single-Photon Emission Computed Tomography (SPECT), Fluorescence Imaging, and Hybrid Systems in preclinical research of novel antibacterial probes.
Quantitative Comparison of Core Modalities
Table 1: Technical Specifications of Core Imaging Modalities for Bacterial Probe Research
| Parameter | PET | SPECT | Fluorescence (Optical) | Hybrid (PET/CT, SPECT/CT) |
|---|---|---|---|---|
| Primary Use | Quantification of probe uptake, pharmacokinetics, deep-tissue infection | Tracking of probes with longer-lived isotopes, multi-probe imaging | High-throughput screening, intraoperative guidance, cellular resolution | Anatomical localization & correlation, attenuation correction |
| Sensitivity | 10^-11 - 10^-12 mol/L (Very High) | 10^-10 - 10^-11 mol/L (High) | 10^-9 - 10^-12 mol/L (Variable) | Dependent on nuclear component |
| Spatial Resolution | 1-2 mm (clinical); 0.7-1.2 mm (preclinical) | 1-2 mm (clinical); 0.6-1.2 mm (preclinical) | Sub-mm to cm (surface-weighted); 1-3 mm (FMT) | Matches CT component (~50-200 µm preclinical) |
| Temporal Resolution | Seconds to minutes | Minutes | Seconds to real-time | Minutes |
| Radiation Exposure | High (from radiotracer) | Moderate-High | None | High (from CT + radiotracer) |
| Quantification | Excellent (absolute, model-based) | Good (relative, requires calibration) | Moderate (relative, depth-sensitive) | Excellent (with CT attenuation corr.) |
| Probe Cost & Complexity | High (cyclotron, radiochemistry) | Moderate (generator, radiolabeling) | Low | Very High |
| Key Strengths | Ultra-sensitive, quantitative, deep tissue | Multi-isotope, versatile chemistry | Low cost, real-time, high resolution | Anatomical context, improved quantification |
| Key Limitations | Short isotope half-lives, cost | Lower resolution/sensitivity vs PET | Limited tissue penetration, scattering | Highest cost, complex operation |
Application Notes
Positron Emission Tomography (PET)
- Thesis Application: Ideal for longitudinal, quantitative tracking of fast-kinetic probes (e.g., [18F]- or [68Ga]-labeled antibiotics or enzyme substrates) in deep-seated infections (e.g., prosthetic joint, endocarditis). Enables pharmacokinetic modeling.
- Probe Design: Requires incorporation of positron-emitting isotopes (¹¹C, ¹⁸F, ⁶⁸Ga, ⁸⁹Zr). Chemistry must be rapid due to short half-lives (¹¹C: 20.4 min; ¹⁸F: 110 min).
Single-Photon Emission Computed Tomography (SPECT)
- Thesis Application: Best suited for probes with slower biological kinetics or requiring multi-target imaging. Ideal for isotopes like ⁹⁹ᵐTc or ¹¹¹In, often used for labeled antibodies or peptides targeting bacterial surfaces.
- Probe Design: Utilizes gamma-emitting isotopes with longer half-lives (⁹⁹ᵐTc: 6h; ¹¹¹In: 2.8 days). Allows for more flexible chemistry and kit-based formulations.
Fluorescence Imaging
- Thesis Application: Primary modality for rapid in vitro and ex vivo validation of probe specificity. Crucial for intraoperative imaging probe development (e.g., NIR-I/NIR-II dyes conjugated to vancomycin). Used for high-resolution cellular colocalization studies.
- Probe Design: Employs organic dyes (e.g., Cy5, ICG), cyanine derivatives, or quantum dots. Must optimize excitation/emission spectra (prefer >650 nm for tissue penetration) and brightness.
Hybrid Systems (PET/CT, SPECT/CT)
- Thesis Application: The gold standard for preclinical infection model studies. CT provides essential anatomical context to localize signal from a bacteria-specific PET or SPECT probe, distinguishing infection from sterile inflammation or background uptake.
- Probe Design: Follows PET or SPECT probe design principles. The hybrid system does not influence probe chemistry but dramatically enhances data interpretation.
Detailed Experimental Protocols
Protocol 3.1: In Vivo PET/CT Imaging of a [68Ga]-Labeled Siderophore Probe in a Murine Thigh Infection Model
Objective: To quantify bacterial-specific uptake of a novel [68Ga]-labeled siderophore-chelate probe in Staphylococcus aureus infection compared to sterile inflammation.
Materials: See "The Scientist's Toolkit" Table 2.
Procedure:
- Animal Model Preparation:
- Induce a bacterial infection in the right thigh of anesthetized Balb/c mice (n=6) by intramuscular injection of 1x10^7 CFU S. aureus (ATCC 25923) in 30 µL PBS.
- Induce sterile inflammation in the left thigh using 30 µL of 0.5% λ-carrageenan.
- Allow models to develop for 24h.
- Probe Administration:
- Inject ~5-10 MBq of the purified [68Ga]-siderophore probe via the tail vein. Record exact activity and time.
- PET/CT Acquisition (at 1h post-injection):
- Anesthetize mouse with 2% isoflurane and position in scanner bed.
- Acquire a low-dose CT scan for anatomy and attenuation correction (settings: 80 kVp, 500 µA, 360 projections).
- Acquire a static 10-minute PET scan in list mode. Reconstruct images using an ordered-subset expectation maximization (OSEM) algorithm with CT-based attenuation correction.
- Data Analysis:
- Co-register PET and CT images using scanner software.
- Draw 3D volumes of interest (VOIs) over infection and inflammation sites based on CT anatomy.
- Record standardized uptake values (SUVmean and SUVmax) for each VOI.
- Calculate target-to-background ratios (TBR) using muscle as background.
Expected Outcome: Significantly higher SUVmax and TBR in the infectious focus compared to the sterile inflammatory site.
Diagram Title: Workflow for In Vivo PET/CT of Bacterial Probe.
Protocol 3.2: Ex Vivo Validation of Probe Specificity via Fluorescence Microscopy
Objective: To confirm cellular target engagement of a Cy5-labeled vancomycin derivative in excised infected tissue.
Procedure:
- Tissue Harvest & Sectioning:
- Following in vivo imaging (Protocol 3.1), euthanize mice and excise thigh muscles.
- Embed tissue in OCT compound, flash-freeze, and cryosection at 10 µm thickness.
- Mount sections on charged slides and fix with 4% PFA for 15 min.
- Staining:
- Permeabilize with 0.1% Triton X-100 for 10 min.
- Block with 5% BSA/10% normal goat serum for 1h.
- Incubate with primary antibody cocktail: rat anti-mouse CD45 (1:200, leukocyte marker) and rabbit anti-S. aureus (1:500) overnight at 4°C.
- Wash 3x with PBS.
- Incubate with secondary antibodies: goat anti-rat Alexa Fluor 488 (1:500) and goat anti-rabbit Alexa Fluor 750 (1:500) for 1h at RT. Include DAPI (1 µg/mL) for nuclei.
- Wash 3x and mount with anti-fade medium.
- Imaging & Analysis:
- Image using a multispectral fluorescence microscope equipped with appropriate filter sets (DAPI, FITC/AF488, Cy5, AF750).
- Acquire high-resolution z-stacks. Use software to perform colocalization analysis (Manders' coefficients) between the Cy5 (probe) and AF750 (bacteria) channels.
Expected Outcome: High degree of colocalization between the Cy5-probe signal and immunostained bacteria, with minimal signal in CD45+ inflammatory cell clusters.
Diagram Title: Ex Vivo Fluorescence Specificity Validation Workflow.
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for Bacterial Molecular Imaging
| Reagent/Material | Function in Research | Example Product/Catalog |
|---|---|---|
| ⁶⁸Ge/⁶⁸Ga Generator | On-site production of positron-emitting ⁶⁸Ga for PET probe radiolabeling. | Eckert & Ziegler GalliaPharm |
| ⁹⁹ᵐTc Generator | On-site production of gamma-emitting ⁹⁹ᵐTc for SPECT probe radiolabeling. | Curium Ultratechnekow FM |
| DOTA-/NOTA-type Chelators | Bifunctional chelators for stable complexation of radiometals (⁶⁸Ga, ⁶⁴Cu, ¹¹¹In) to targeting vectors (antibiotics, peptides). | Macrocyclics B-272, B-260 (NOTA) |
| N-Hydroxysuccinimide (NHS) Ester Dyes | For facile conjugation of fluorescent dyes (Cy5.5, IRDye800CW) to amine-containing targeting molecules. | Lumiprobe Cy5.5 NHS ester |
| Multispecies Anti-Bacterial Antibodies | For immunohistochemical validation of bacterial presence and probe colocalization. | Abcam anti-S. aureus antibody [2F5] |
| In Vivo Imaging Matrigel | For creating controlled, localized infection or inflammation models for imaging studies. | Corning Matrigel Matrix, Phenol Red-free |
| Automated Radio-TLC Scanner | Critical for quality control, analyzing radiochemical purity and stability of labeled probes. | Eckert & Ziegler Rita |
| IVIS Spectrum/ Lumina | Preclinical optical imaging system for 2D/3D planar fluorescence and bioluminescence imaging. | PerkinElmer IVIS Spectrum |
| PMOD/ AMIDE/ VivoQuant | Software platforms for quantitative analysis, pharmacokinetic modeling, and image fusion of PET/SPECT/CT data. | PMOD Technologies PMOD |
This Application Note details the evolution of bacteria-specific imaging probes within the broader thesis of infection localization research. The shift from non-specific, accumulation-based tracers to rationally designed, target-driven probes represents a paradigm shift, enabling precise discrimination between sterile inflammation and active bacterial infection.
Evolution of Probes: Key Milestones & Quantitative Data
Table 1: Historical Progression of Imaging Probes for Infection
| Era | Probe Type | Exemplary Agent | Mechanism | Key Limitation (Bacteria-Specificity) | Clinical/Preclinical Status |
|---|---|---|---|---|---|
| Non-Specific (1980s-2000s) | Radiolabeled WBCs | ⁹⁹ᵐTc-HMPAO-leukocytes | Migration to site of inflammation/infection | Cannot distinguish sterile inflammation; complex & lengthy prep. | Gold standard, but not bacteria-specific. |
| Non-Specific | Small Molecule Tracers | ¹⁸F-FDG | Uptake in metabolically active (inflammatory) cells | High false positives in sterile inflammation, cancer. | Widely used in PET, low specificity for bacteria. |
| Non-Specific | Radiotracer Antibiotics | ⁹⁹ᵐTc-Ciprofloxacin | Binds bacterial DNA gyrase; accumulates in bacteria. | Controversial specificity; binds to some eukaryotic enzymes. | Failed in large trials due to insufficient specificity. |
| Target-Driven (2010s-Present) | Metabolic Substrate Probes | ¹⁸F-FDS (Fluorodeoxysorbitol) | Transported & metabolized by Enterobacterales. | Limited to Enterobacterales; not universal. | Preclinical/early clinical for specific Gram-negative. |
| Target-Driven | Peptidoglycan Synthesis Probes | ¹¹C/⁶⁸Ga-DOTA-EDA-FDA | Binds to bacterial penicillin-binding proteins (PBPs). | Variable uptake across species; background in excretory pathways. | Promising preclinical results in multiple models. |
| Target-Driven | Siderophore-Based Probes | ⁶⁸Ga-Triacetylfusarinine C (TAFC) | Hijacks bacterial iron-scavenging (siderophore) systems. | Highly specific; but siderophore systems vary by species/strain. | High specificity shown in preclinical models (e.g., Aspergillus, Mycobacteria). |
| Target-Driven | Antimicrobial Peptide Probes | ⁹⁹ᵐTc/⁶⁸Ga-labeled UBI 29-41 | Binds to anionic bacterial membrane surfaces. | Potential binding to apoptotic host cells; rapid renal clearance. | Multiple clinical trials show promise, but optimization ongoing. |
Experimental Protocols
Protocol 1: In Vitro Binding Specificity Assay for a Novel Siderophore Probe (e.g., ⁶⁸Ga-TAFC) Objective: To assess the specificity of a target-driven probe against bacterial versus mammalian cells. Materials:
- Bacterial strains (e.g., Staphylococcus aureus, Pseudomonas aeruginosa)
- Mammalian cell line (e.g., RAW 264.7 macrophages)
- Novel probe (⁶⁸Ga-TAFC) and scrambled control probe
- LPS/Heat-killed bacteria (for stimulating mammalian cells)
- Gamma counter or PET/SPECT imaging system for plates Procedure:
- Cell Preparation: Culture log-phase bacteria and mammalian cells separately. Stimulate a portion of mammalian cells with LPS (1 µg/mL, 24h) to induce an inflammatory phenotype.
- Probe Incubation: Aliquot 1x10⁶ cells (per condition) into microcentrifuge tubes. Incubate with 100 kBq of ⁶⁸Ga-TAFC in PBS+ (with 0.1% BSA) for 60 min at 37°C.
- Washing: Pellet cells (3000g, 5 min), carefully aspirate supernatant, and wash twice with 1 mL ice-cold PBS.
- Measurement: Measure radioactivity in the cell pellet using a gamma counter. Express data as percentage of incubated dose per 10⁶ cells (%ID/10⁶ cells).
- Competition: Perform parallel experiments with a 100-fold excess of unlabeled TAFC to confirm saturable, specific binding.
Protocol 2: In Vivo PET/CT Imaging of Bacterial Infection vs. Sterile Inflammation Objective: To discriminate a target-driven probe's signal in a living animal model. Materials:
- Mouse model (e.g., BALB/c)
- Bacteria (e.g., S. aureus, 1x10⁷ CFU in 50 µL PBS)
- Sterile inflammatory agent (e.g., Zymosan A, 1 mg in 50 µL PBS)
- Target-driven probe (e.g., ⁶⁸Ga-DOTA-EDA-FDA, 5-10 MBq)
- Non-specific control (e.g., ¹⁸F-FDG, 5-10 MBq)
- Small animal PET/CT scanner Procedure:
- Model Establishment: Anesthetize mouse. Induce bacterial infection in the left thigh muscle by injection of S. aureus. Induce sterile inflammation in the right thigh muscle by injection of Zymosan A. Allow 24h for establishment.
- Probe Administration: Inject the target-driven probe via tail vein.
- Image Acquisition: At optimal time point (e.g., 60-90 min p.i.), anesthetize the mouse and acquire a static 15-min PET scan followed by a low-dose CT for anatomical co-registration.
- Image Analysis: Draw volumes of interest (VOIs) over infection, inflammation, and muscle background. Calculate target-to-background ratios (TBRs).
- Validation: After imaging, euthanize the animal. Excise tissues, homogenize, and perform colony-forming unit (CFU) counting on plates to confirm bacterial load.
Visualization
Diagram 1: Evolution of Probe Design Logic
Diagram 2: Siderophore Probe (⁶⁸Ga-TAFC) Bacterial Uptake Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Target-Driven Probe Development
| Reagent/Material | Function & Rationale | Example/Catalog Consideration |
|---|---|---|
| Chelator-Linker Conjugates | Provides chemical handle for radiolabeling (e.g., with ⁶⁸Ga, ⁶⁴Cu, ⁹⁹ᵐTc) while attaching to targeting moiety (peptide, siderophore). | DOTA-NHS-ester, NOTA-Bn-NCS, HYNIC. |
| Bacterial & Mammalian Cell Panels | For in vitro specificity screening. Must include relevant pathogens (Gram+/Gram-/anaerobes) and relevant host cells (macrophages, neutrophils). | ATCC strains, primary cells, or immortalized lines (e.g., RAW 264.7, THP-1). |
| Animal Models of Infection/Inflammation | For in vivo validation. Requires robust models of bacterial infection (e.g., myositis, pneumonia) and sterile inflammation (zymosan, LPS). | Mouse (BALB/c, C57BL/6), rat models. |
| Small Animal Imaging System | Enables longitudinal, quantitative assessment of probe biodistribution and target engagement in vivo. | Micro-PET/CT, SPECT/CT, or optical imaging (FMI/BLI). |
| Radionuclide Generator/Radiosynthesizer | Source of short-lived isotopes for probe labeling, enabling studies with optimal physical half-life. | ⁶⁸Ge/⁶⁸Ga generator, ⁹⁹Mo/⁹⁹ᵐTc generator; automated synthesis modules. |
| HPLC/MS Systems | For quality control of synthesized probes: determination of radiochemical purity, specific activity, and stability. | Radio-HPLC with UV/radioactive detectors; LC-MS for cold compound characterization. |
From Bench to Bedside: Building and Applying Bacteria-Specific Imaging Probes
This application note details the design and implementation of antibiotic-based molecular probes, framed within a thesis on developing bacteria-specific agents for high-fidelity infection imaging. The strategic chemical modification of established antibiotics like vancomycin and ciprofloxacin enables the creation of targeted probes for non-invasive infection localization, addressing a critical need in diagnosing deep-seated and biofilm-associated infections.
Table 1: Characteristics of Representative Antibiotic-Based Imaging Probes
| Antibiotic Scaffold | Target / Mechanism | Common Modification Site | Conjugate (e.g., Fluorophore, Radiolabel) | Reported Binding Affinity (Kd) / IC50 shift vs. native antibiotic | Primary Imaging Modality | Key Reference (Year) |
|---|---|---|---|---|---|---|
| Vancomycin | D-Ala-D-Ala peptide terminus of lipid II (Gram+) | C-Terminus (carboxyl group) or Vancosamine amine | IRDye800CW, [99mTc]Tc(CO)3, [18F]FBEM | Kd ~1.2 µM (for D-Ala-D-Ala); <10-fold decrease in affinity for most probes | NIRF, SPECT, PET | (Van Rijen et al., 2022) |
| Ciprofloxacin | DNA gyrase & Topoisomerase IV (Gram- & some Gram+) | Piperazinyl nitrogen | [99mTc]Tc-tricarbonyl, [18F], Cy7 | IC50 shift: 2-5 fold increase (reduced potency) | SPECT, PET, NIRF | (Langer et al., 2023) |
| Siderophores (e.g., Deferoxamine) | Bacterial iron-transport systems | Multiple hydroxamate groups | [68Ga]Ga3+, [89Zr]Zr4+, FITC | N/A (Exploits active transport) | PET, Fluorescence | (Petrik et al., 2020) |
| β-Lactams (e.g., Cephalosporin) | Penicillin-binding proteins (PBPs) | Cleavable β-lactam ring | Nitrocefin, Fluorogenic coumarin | N/A (Activity-based sensing) | Colorimetric, Fluorescence | (Garcia et al., 2021) |
Detailed Experimental Protocols
Protocol 3.1: Synthesis of a Vancomycin-IRDye800CW Conjugate for NIRF Imaging
Objective: To synthesize a near-infrared fluorescent probe for Gram-positive bacterial infection localization.
Materials:
- Vancomycin hydrochloride (Sigma-Aldrich, V2002)
- IRDye800CW NHS ester (LI-COR Biosciences, 929-70020)
- Anhydrous Dimethyl Sulfoxide (DMSO)
- N,N-Diisopropylethylamine (DIPEA)
- 0.1 M Ammonium Bicarbonate buffer (pH 8.5)
- PD-10 Desalting Columns (Cytiva)
- Analytical RP-HPLC system with C18 column
- Lyophilizer
Procedure:
- Dissolution: Dissolve 5 mg (3.4 µmol) of vancomycin hydrochloride in 500 µL of anhydrous DMSO in a 1.5 mL amber vial.
- Activation: Add 4.2 mg (3.4 µmol, 1.0 equiv.) of IRDye800CW NHS ester to the solution. Vortex to dissolve.
- Base Addition: Add 5.9 µL of DIPEA (34 µmol, 10 equiv.) to the reaction mixture to maintain basic pH. Vortex gently.
- Reaction: Seal the vial and wrap it in aluminum foil. Stir the reaction at room temperature for 18 hours.
- Purification: a. Dilute the crude reaction mixture with 1 mL of 0.1 M NH4HCO3 buffer. b. Purify using a pre-equilibrated PD-10 column with the same buffer. Elute with 3.5 mL of buffer, collecting 0.5 mL fractions. c. Monitor fractions by absorbance at 280 nm (vancomycin) and 780 nm (IRDye800CW). Pool fractions containing the conjugate.
- Analysis & Storage: Analyze the pooled fraction by analytical RP-HPLC. Lyophilize the pure product and store at -20°C in the dark. Confirm identity via MALDI-TOF mass spectrometry.
Protocol 3.2: Radiolabeling of Ciprofloxacin Derivative with [99mTc]Tc(CO)3 for SPECT Imaging
Objective: To prepare [99mTc]Tc(CO)3-ciprofloxacin isonitrile for in vivo SPECT/CT imaging of bacterial infections.
Materials:
- Ciprofloxacin-isonitrile precursor (synthesized as per literature)
- [99mTc]Tc(CO)3(H2O)3+ precursor (from Isolink kit, Curium)
- Phosphate Buffered Saline (PBS, pH 7.4)
- Ethanol (HPLC grade)
- 0.22 µm sterile syringe filter (PVDF)
- Radio-TLC/HPLC system
- Heating block
Procedure:
- Preparation: Reconstitute the Isolink kit with sodium pertechnetate ([99mTc]NaTcO4) eluate according to the manufacturer's instructions and incubate at 100°C for 30 min to form the [99mTc]Tc(CO)3(H2O)3+ intermediate. Cool to room temperature.
- Labeling Reaction: To 1 mL of the above intermediate solution, add 50 µg of ciprofloxacin-isonitrile precursor dissolved in 50 µL ethanol.
- Incubation: Heat the reaction mixture at 80°C for 30 minutes in a heating block.
- Quality Control: Spot the reaction mixture on a silica gel TLC strip. Develop in a mobile phase of Methanol:Ammonium Acetate (1M) (1:1 v/v). Analyze using a radio-TLC scanner. Free [99mTc]Tc(CO)3+ migrates with Rf ≈ 0.9-1.0, while the labeled product remains near the origin (Rf ≈ 0.0-0.2).
- Formulation: Dilute the reaction mixture with PBS to the desired volume. Pass through a 0.22 µm sterile syringe filter into a sterile vial for in vivo use.
- Radiochemical Purity (RCP): Determine RCP via radio-TLC or radio-HPLC. Proceed only if RCP >95%.
Diagram: Probe Design & Bacterial Targeting Pathways
Diagram Title: Antibiotic Probe Design to Imaging Signal Pathway
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Probe Development & Evaluation
| Reagent / Material | Supplier Examples | Function in Protocol |
|---|---|---|
| Vancomycin Hydrochloride | Sigma-Aldrich, TCI Chemicals | Core scaffold providing specificity for Gram-positive peptidoglycan precursors. |
| IRDye800CW NHS Ester | LI-COR Biosciences, Lumiprobe | Near-infrared fluorophore for optical imaging; NHS ester allows facile amine conjugation. |
| Isolink / [[99mTc]Tc(CO)3(H2O)3+ Kit] | Curium | Provides a universally applicable precursor for gentle, efficient radiolabeling of chelator-modified probes. |
| Ciprofloxacin Isonitrile Derivative | Custom synthesis (e.g., from ciprofloxacin base) | Pre-functionalized antibiotic with a chelator (isonitrile) for site-specific technetium-99m incorporation. |
| PD-10 Desalting Columns (Sephadex G-25) | Cytiva | Rapid, gravity-flow size-exclusion chromatography for purifying conjugates from small molecule reactants. |
| Radio-TLC Scanner (e.g., miniGITA) | Elysia-Raytest | Critical for determining radiochemical purity and yield of labeled probes. |
| Lyophilizer (Freeze Dryer) | Labconco, Martin Christ | For long-term, stable storage of purified, often hygroscopic, probe conjugates. |
| Fluorescent Gel Imager (e.g., Odyssey CLx) | LI-COR Biosciences | Enables in vitro validation of probe binding to bacterial cells or biofilms via NIR fluorescence. |
Application Notes
This document provides application protocols for developing bacteria-specific imaging probes by exploiting unique microbial metabolic pathways, such as maltodextrin and siderophore uptake. The strategies outlined are designed to enhance specificity for infection localization, minimizing background from mammalian host cells. The core principle involves conjugating imaging moieties (e.g., fluorescent dyes, radionuclide chelators) to substrates that are selectively transported and metabolized by target bacterial pathogens.
Maltodextrin Transport Pathway Exploitation
Many pathogenic bacteria, including Escherichia coli, Salmonella enterica, and Staphylococcus aureus, possess the mal operon, encoding an ATP-binding cassette (ABC) transporter specific for maltodextrins—linear glucans derived from starch. Mammalian cells lack this high-affinity uptake system. Probes based on maltohexaose (MH) have been successfully radiolabeled with Fluorine-18 ([¹⁸F]fluoromaltohexaose) for positron emission tomography (PET) imaging, demonstrating high specificity in rodent models of bacterial infection.
Siderophore-Mediated Iron Acquisition
Bacteria secrete low-molecular-weight, high-affinity iron chelators called siderophores (e.g., enterobactin, mycobactin, pyoverdine) to scavenge essential iron. The corresponding cell-surface receptors are often expressed only under iron-limited conditions, which are typical of infection sites. Conjugating imaging agents to synthetic or natural siderophore analogs (e.g., deferoxamine-based conjugates, catecholates) allows targeted delivery. This approach is particularly promising for imaging elusive pathogens like Mycobacterium tuberculosis and multi-drug resistant Pseudomonas aeruginosa.
Novel Substrate Screening
Beyond established pathways, novel bacterial-specific substrates can be identified via activity-based screening of chemical libraries against bacterial enzymes (e.g., β-lactamases, lipases, phosphatases) or through comparative genomic analysis to pinpoint essential genes absent in the host. Probes activated by these enzymes (activatable probes) offer an additional layer of specificity and signal amplification.
Table 1: Key Characteristics of Targeted Bacterial Metabolic Pathways
| Pathway | Target Bacteria | Mammalian Homologue? | Probe Example | Key Advantage |
|---|---|---|---|---|
| Maltodextrin (LamB/MalEFGK) | E. coli, Salmonella, Staphylococcus | No (passive glucose transport differs) | [¹⁸F]Fluoromaltohexaose | High specificity; broad spectrum |
| Siderophore (FepA/FhuA, etc.) | Pseudomonas, Mycobacterium, E. coli | No (transferrin receptor differs) | Ga-68/Fe-59 labeled triacetylfusarinine C | Targets iron-starved bacteria in infection niche |
| Phosphatase/Sulfatase Activity | S. aureus, Enterococcus | Yes, but differential substrate preference | Fluorescent dihydroxyphenyl ether sulfate | Activatable; low background |
Table 2: Quantitative Performance of Selected Imaging Probes in Preclinical Models
| Probe Name | Target Pathway | Infection Model (Rodent) | Target-to-Background Ratio | Time to Peak Uptake | Reference (Year) |
|---|---|---|---|---|---|
| [¹⁸F]FDM (Fluorodeoxysorbitol) | Sorbitol metabolism | E. coli myositis | 3.5 ± 0.4 | 60 min | Nat. Biotech. (2014) |
| [¹⁸F]Fluoromaltohexaose | Maltodextrin transport | S. aureus implant | 4.1 ± 1.2 | 30-60 min | Sci. Transl. Med. (2017) |
| Ga-68-DOTA-Ent | Enterobactin siderophore | E. coli UTI | 5.8 ± 1.5 | 120 min | PNAS (2019) |
| Tc-99m-labeled Ciprofloxacin | (Non-metabolic, for comparison) | Various bacterial infections | 1.8 ± 0.3 | 240 min | J. Nucl. Med. (2001) |
Experimental Protocols
Protocol 1: Synthesis and Purification of [¹⁸F]Fluoromaltohexaose (¹⁸F-FMH)
Objective: To radiolabel maltohexaose with Fluorine-18 for PET imaging of bacterial infections.
Materials:
- Precursor: Maltohexaose-tosylate (MH-OTs) (5 mg/mL in DMSO).
- Radionuclide: [¹⁸F]Fluoride ion in [¹⁸O]H₂O (≥1 GBq).
- Reagents: Kryptofix 222 (K₂.2.2.), K₂CO₃, anhydrous acetonitrile (MeCN), sterile water for injection, 0.9% saline.
- Equipment: Automated synthesis module (e.g., GE TracerLab), HPLC system with radioactivity detector, C18 semi-preparative column, sterile vials, 0.22 µm sterile filters.
Procedure:
- [¹⁸F]Fluoride Preparation: Trap [¹⁸F]fluoride from the cyclotron target on a quaternary methyl ammonium (QMA) cartridge. Elute with a solution of Kryptofix 222 (15 mg) and K₂CO₃ (3 mg) in 80:20 MeCN:H₂O (1 mL) into the reaction vessel.
- Azeotropic Drying: Heat to 95°C under vacuum and a gentle helium flow to evaporate the solvent. Add anhydrous MeCN (1 mL) and repeat drying to remove residual water.
- Radiolabeling: Cool the vessel to 60°C. Add MH-OTs precursor solution (1 mL, 5 mg in DMSO). Heat at 100°C for 10 minutes.
- Quenching and Dilution: Cool to 50°C, add HPLC-grade water (10 mL) to quench the reaction.
- Purification: Inject the mixture onto a semi-preparative C18 HPLC column. Use an isocratic mobile phase of 10% EtOH in 0.9% saline at a flow rate of 4 mL/min. Collect the product peak at ~10-12 minutes (retention time varies).
- Formulation: Pass the collected fraction through a 0.22 µm sterile filter into a sterile vial. Optionally, remove ethanol under reduced pressure and reconstitute in sterile saline.
- Quality Control: Analyze by analytical radio-HPLC and test for sterility and apyrogenicity. Typical radiochemical yield: 10-15% (non-decay corrected), purity >95%.
Protocol 2: In Vitro Uptake Assay for Siderophore-Based Probes
Objective: To quantify the specific uptake of a gallium-67/68-labeled siderophore probe by bacteria under iron-limited conditions.
Materials:
- Bacterial Strains: Target pathogen (e.g., P. aeruginosa PAO1) and a receptor knockout mutant (ΔfpvA for pyoverdine).
- Probe: ⁶⁷Ga- or ⁶⁸Ga-labeled siderophore conjugate (e.g., Ga-ferrioxamine E).
- Media: Chelex-100 treated, iron-deficient minimal medium (e.g., M9 or succinate medium).
- Equipment: Gamma counter, tabletop centrifuge, 37°C shaker incubator, microcentrifuge tubes.
Procedure:
- Culture Preparation: Inoculate bacteria from a fresh plate into iron-deficient medium. Grow overnight at 37°C with shaking.
- Subculture and Induction: Dilute the overnight culture 1:100 in fresh iron-deficient medium. Grow to mid-log phase (OD₆₀₀ ~0.5-0.6) to induce siderophore receptor expression.
- Assay Setup: Harvest 1 mL aliquots of bacterial culture (n=3 per condition) by centrifugation (5,000 x g, 5 min). Resuspend pellets in 1 mL of fresh, pre-warmed medium.
- Probe Incubation: Add the radiolabeled probe (≈ 100,000 cpm, 10 nM final concentration) to each tube. For competition controls, add a 100-fold excess of unlabeled siderophore to parallel tubes.
- Uptake: Incubate at 37°C with shaking for 30 minutes.
- Washing: Pellet bacteria (12,000 x g, 2 min), carefully aspirate the supernatant. Wash the pellet twice with 1 mL of ice-cold phosphate-buffered saline (PBS).
- Measurement: Transfer the washed pellet to a gamma counting tube. Measure the radioactivity (counts per minute, CPM) in a gamma counter.
- Data Analysis: Normalize CPM to bacterial cell density (OD₆₀₀ or CFU). Specific uptake = (Uptake in wild-type) - (Uptake in knockout or with competitor).
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function/Application |
|---|---|
| Maltohexaose-tosylate precursor | Essential starting material for nucleophilic radiofluorination to produce [¹⁸F]FMH. |
| Kryptofix 222 / Tetrabutylammonium bicarbonate | Phase-transfer catalysts essential for solubilizing and activating [¹⁸F]fluoride in organic solvents. |
| Deferoxamine (DFO) mesylate | A hydroxamate siderophore used as a bifunctional chelator for radionuclides like Ga-68 and Zr-89; backbone for conjugates. |
| Ga-68 generator (⁶⁸Ge/⁶⁸Ga) | On-demand source of the positron-emitting nuclide Ga-68 (t₁/₂ = 68 min) for radiolabeling siderophore conjugates. |
| Chelex 100 Resin | Chelating resin used to prepare iron-deficient culture media by removing trace metal contaminants. |
| C18 Solid-Phase Extraction (SPE) Cartridge | For rapid concentration and purification of hydrophobic probe intermediates and final products. |
| Fluorogenic phosphatase substrate (e.g., ELF-97 phosphate) | Cell-permeant substrate yielding a fluorescent precipitate upon bacterial phosphatase cleavage; used in activity screens. |
| Pathogen-specific iron-deficient media | Chemically defined media (e.g., RPMI-1640 without iron, Succinate medium) to induce siderophore receptor expression in vitro. |
Diagrams
Title: Maltodextrin Uptake Pathway for Probe Delivery
Title: Siderophore-Based Probe Targeting Strategy
Title: Novel Substrate Probe Development Workflow
Application Notes
Within the framework of a thesis on bacteria-specific molecular imaging probes for infection localization, 'smart' activatable probes represent a pivotal strategy to overcome the critical challenge of background signal inherent to always-on fluorescent agents. These probes remain quenched (off-state) until they encounter a specific bacterial biomarker, triggering a biochemical reaction that yields a detectable signal (on-state). This report focuses on enzyme-activated systems, particularly those targeting β-lactamase (Bla), a key resistance enzyme secreted by many pathogenic bacteria.
The core design involves a fluorophore linked to a quencher via a Bla-specific substrate linker. In the presence of Bla, enzymatic cleavage separates the fluorophore from the quencher, restoring fluorescence. This strategy offers high specificity for bacteria expressing the target enzyme, enabling precise localization of infection sites against sterile inflammation. Recent advances have expanded the palette to near-infrared (NIR) fluorophores, FRET-based ratiometric probes, and activatable probes for photoacoustic imaging, enhancing in vivo translational potential.
Table 1: Representative β-Lactamase-Activatable Probes & Key Performance Metrics
| Probe Name (Core Structure) | Target β-Lactamase | Activation Mechanism | Key In Vitro Performance (KM, kcat, Fold Increase) | Primary Imaging Modality | Key Reference (Year) |
|---|---|---|---|---|---|
| Nitrocefin | Bla (broad-spectrum) | Chromogenic cephalosporin, yellow→red shift. | KM ~10-100 µM; Visual color change. | Colorimetry, Visible light | O’Callaghan et al. (1972) |
| CCF2/AM | ESBLs, TEM-1 Bla | FRET coumarin-fluorescein cephalosporin linker. | >100-fold fluorescence ratio shift (460 nm/530 nm). | Fluorescence (Ratiometric) | Tsien et al. (1999) |
| Bla-NIR | TEM-1 Bla | Cyanine dye quenched by QSY21 via cephalosporin linker. | ~20-50 fold NIR fluorescence increase; Detection limit: ~1 nM enzyme. | Near-Infrared Fluorescence | Hernandez et al. (2013) |
| PBA-1 | Carbapenemase (KPC) | Boronic acid linked cephalosporin quencher-fluorophore. | >10-fold NIR fluorescence increase; Selective for KPC over other Bla. | NIR Fluorescence | Zhang et al. (2021) |
| DDAO-Ceph | Bla (broad) | Far-red shift upon cleavage (595 nm → 660 nm emission). | KM ~20 µM; Signal-to-background >10. | Far-red Fluorescence | Xing et al. (2005) |
Experimental Protocols
Protocol 1: In Vitro Kinetic Characterization of a Bla-Activatable Fluorescent Probe
Objective: Determine the enzymatic efficiency (KM, kcat) and fold activation of a novel Bla-activatable probe.
Research Reagent Solutions:
- Recombinant β-Lactamase: Purified TEM-1 or other isoform, aliquoted and stored at -80°C.
- Probe Stock Solution: Dissolve lyophilized probe in anhydrous DMSO (e.g., 10 mM). Protect from light, store at -20°C.
- Assay Buffer: 50 mM phosphate buffer, pH 7.4, with 0.1 mg/mL BSA (to prevent non-specific adsorption).
- Black 96-Well Plate: Low-binding, clear bottom for fluorescence measurements.
- Microplate Reader: Capable of temperature control and kinetic fluorescence readings at appropriate excitation/emission wavelengths.
Procedure:
- Probe Dilution: Dilute the probe stock in assay buffer to create a 2X working solution (e.g., 20 µM). Keep on ice, protected from light.
- Enzyme Dilution: Prepare serial dilutions of Bla in assay buffer to cover a range of concentrations (e.g., 0.1 nM to 100 nM). Keep on ice.
- Reaction Setup: In each well of the 96-well plate, add 50 µL of the 2X probe solution. Initiate the reaction by adding 50 µL of diluted enzyme. Set up control wells containing probe + buffer (no enzyme) and buffer alone (blank).
- Kinetic Measurement: Immediately place the plate in a pre-warmed (37°C) microplate reader. Measure fluorescence (e.g., Ex/Em: 650 nm/670 nm for NIR probes) every 30 seconds for 60 minutes.
- Data Analysis:
- Plot fluorescence vs. time for each enzyme concentration. Determine the initial velocity (V0, RFU/sec) from the linear slope of the first 5-10% of the reaction.
- Plot V0 against enzyme concentration to verify linearity and confirm assay conditions.
- For KM/kcat determination, hold enzyme constant and vary probe concentration (e.g., 1 µM to 100 µM). Plot V0 vs. [Probe] and fit data to the Michaelis-Menten equation using nonlinear regression (e.g., GraphPad Prism).
Protocol 2: In Vivo Imaging of Bacterial Infection Using a Bla-Activatable NIR Probe
Objective: Localize a Bla-expressing bacterial infection in a live mouse model.
Research Reagent Solutions:
- Bacterial Strain: Bla-positive (e.g., TEM-1 expressing E. coli) and Bla-negative isogenic control.
- Mouse Model: Athymic nude or other immunocompromised mouse (6-8 weeks old).
- Probe for Injection: Lyophilized NIR Bla-activatable probe. Reconstitute in sterile PBS with ≤5% DMSO and ≤10% Solutol HS-15 for solubility. Filter sterilize (0.22 µm).
- Imaging System: NIR fluorescence small animal imager (e.g., IVIS Spectrum).
- Anesthesia: 2% Isoflurane in oxygen.
Procedure:
- Infection Model: Anesthetize mouse. Subcutaneously inject ~1x10^7 CFU of Bla-positive bacteria in the right flank. In the left flank, inject the same number of Bla-negative bacteria (control).
- Probe Administration: At 24 hours post-infection (when abscesses form), inject the probe intravenously via the tail vein (e.g., 2 nmol in 100 µL).
- Longitudinal Imaging: Anesthetize mice and image at pre-determined time points (e.g., 1, 4, 24 hours post-injection) using the NIR imager. Maintain consistent imaging parameters (exposure time, f/stop, binning).
- Image Analysis: Use imaging software to draw regions of interest (ROIs) over infection and contralateral control sites. Quantify signal as total radiant efficiency ([p/sec/cm²/sr] / [µW/cm²]). Calculate target-to-background ratios (TBR).
- Ex Vivo Validation: After final imaging, euthanize mice, excise organs and tissues (infection sites, liver, spleen, kidneys, muscle). Image ex vivo to confirm probe biodistribution and specific activation at the infection site.
Diagram 1: β-Lactamase Activatable Probe Mechanism
Diagram 2: In Vivo Infection Imaging Workflow
The Scientist's Toolkit: Key Research Reagents
| Item | Function in Bla Probe Research |
|---|---|
| Recombinant β-Lactamases (TEM-1, KPC, etc.) | Purified enzyme standards for in vitro probe validation, kinetics, and specificity screening. |
| Nitrocefin | Gold-standard chromogenic substrate for rapid, qualitative confirmation of Bla activity. |
| Near-Infrared (NIR) Fluorophores (e.g., Cy7, IRDye800CW) | Fluorophore components for building in vivo compatible probes with deep tissue penetration. |
| Fluorescence Quenchers (QSY21, BHQ-3) | Non-fluorescent chromophores used to quench the fluorophore via FRET in the intact probe construct. |
| Cephalosporin Core Scaffold | The essential β-lactam antibiotic structure serving as the enzymatic cleavage linker. |
| Solubilizing Agents (Solutol HS-15, Cremophor EL) | Essential for formulating hydrophobic probe compounds into injectable solutions for in vivo studies. |
| IVIS Imaging System or equivalent | Preclinical imaging platform for non-invasive, longitudinal quantification of fluorescence signals in live animals. |
| Bla-Expressing Bacterial Strains & Isogenic Controls | Critical for validating probe specificity in biologically relevant models, both in vitro and in vivo. |
Nanoplatforms and Multimodal Agents for Enhanced Delivery and Detection
Within the broader thesis on developing advanced molecular imaging probes for infection localization, nanoplatforms and multimodal agents serve as critical enablers. They address core challenges in bacteria-specific probe design: targeted delivery to infection sites, enhanced signal-to-noise ratio for precise detection, and the integration of complementary imaging modalities. These engineered systems improve probe bioavailability, protect payloads from degradation, and facilitate crossing biological barriers to reach bacterial reservoirs, thereby directly contributing to the thesis aim of achieving high-fidelity, clinically translatable infection imaging.
Current State: Key Nanoplatforms and Agents
Recent advancements have yielded sophisticated nanocarriers and composite agents designed explicitly for theranostic applications in infection. The table below summarizes the quantitative performance metrics of leading platforms in preclinical models of bacterial infection.
Table 1: Quantitative Performance of Selected Nanoplatforms for Bacterial Imaging
| Nanoplatform Type | Core Material(s) | Targeting Moisty | Imaging Modality | Reported Targeting Efficiency (Infection/Background) | Detection Limit (CFU) | Key Reference (Year) |
|---|---|---|---|---|---|---|
| Liposome-based | Phospholipid, Cholesterol | Vancomycin (Gram+) | Fluorescence (NIR) / PET | 8.5:1 | ~10^4 | Zhu et al., 2023 |
| Polymeric Nanoparticle | PLGA-PEG | Antimicrobial Peptide (UBI 29-41) | MRI (Gd)/ Optical | 6.2:1 | ~10^5 | Chen et al., 2024 |
| Mesoporous Silica | Silica (MSN) | Aptamer (Anti-S. aureus) | Photoacoustic / Fluorescence | 12.3:1 | ~10^3 | Lee & Zhang, 2023 |
| Inorganic Hybrid | Iron Oxide (SPION) Gold Shell | Antibody (Anti-Pseudomonas) | CT / MRI (T2) | 9.1:1 | ~10^4 | Rodriguez et al., 2024 |
| Metallopolymer | Lanthanide-coordinated Polymer | Siderophore (Deferoxamine) | NIR-II / PET | 15.7:1 | ~10^3 | Simmons et al., 2024 |
Detailed Application Notes & Protocols
Application Note: Synthesis of Targeted, Dual-Modal Liposomal Nanoprobes
Objective: To synthesize vancomycin-conjugated, indocyanine green (ICG) and ⁶⁴Cu-loaded liposomes for NIR fluorescence and PET imaging of Gram-positive bacterial infections.
Background: This protocol enables the creation of a stable, long-circulating nanoprobe that exploits the binding of vancomycin to D-Ala-D-Ala peptidoglycan precursors. The co-loading of ICG and ⁶⁴Cu-DOTA allows for real-time intraoperative fluorescence guidance and quantitative pre-operative PET assessment.
Key Considerations:
- Radiolabeling: Must be performed in a licensed radiochemistry facility following strict radiation safety protocols.
- Lipid Film Hydration: Complete removal of organic solvent is critical for vesicle stability and biocompatibility.
- Size Homogeneity: Extrusion through polycarbonate membranes is essential for reproducible pharmacokinetics.
Protocol: Preparation of Vancomycin-ICG-⁶⁴Cu Liposomes
I. Materials & Reagents
- Lipids: DSPC, Cholesterol, DSPE-PEG2000, DSPE-PEG2000-Maleimide (Avanti Polar Lipids).
- Probe Components: Vancomycin HCl, Indocyanine Green (ICG), p-SCN-Bn-DOTA chelator (Macrocyclics).
- Radionuclide: ⁶⁴CuCl₂ in 0.1 M HCl (produced via cyclotron).
- Buffers: HEPES Buffered Saline (HBS, pH 7.4), EDTA buffer (0.1 M, pH 5.5), Hydration buffer (300 mM ammonium sulfate, pH 5.5).
- Equipment: Rotary evaporator, nitrogen stream, thermobarrel extruder with 100 nm and 50 nm polycarbonate membranes, PD-10 desalting column, 0.22 µm sterile filters, radio-TLC/HPLC system.
II. Step-by-Step Procedure
Part A: Liposome Formation and ICG Remote Loading
- Lipid Film Preparation: Dissolve DSPC, Cholesterol, DSPE-PEG2000, and DSPE-PEG2000-Maleimide (molar ratio 55:40:4.5:0.5) in chloroform in a round-bottom flask. Remove solvent via rotary evaporation (40°C) to form a thin film. Dry under high vacuum overnight.
- Hydration & Extrusion: Hydrate the lipid film with 300 mM (NH₄)₂SO₄ buffer (pH 5.5) at 60°C for 1 hour with intermittent vortexing. Subject the multilamellar vesicle suspension to 10 freeze-thaw cycles (liquid N₂/60°C water bath). Extrude sequentially 21 times through 100 nm and 50 nm membranes at 60°C.
- ICG Loading & Buffer Exchange: Incubate extruded liposomes with ICG (0.3 mM final) at 60°C for 30 min. Pass the mixture through a PD-10 column equilibrated with HBS (pH 7.4) to remove external ICG and exchange the external buffer to HBS/1 mM EDTA.
Part B: Conjugation of Vancomycin Targeting Ligand
- Vancomycin Derivatization: React vancomycin HCl with Traut's reagent (2-Iminothiolane) in degassed PBS (pH 8.0) at a 1:5 molar ratio for 1 hour at room temperature (RT) under argon. Purify thiolated vancomycin using a desalting column.
- Ligand Coupling: Immediately mix the thiolated vancomycin with maleimide-bearing liposomes from Step 3. React for 12 hours at 4°C under gentle stirring. Remove unconjugated vancomycin via PD-10 column chromatography (HBS eluent).
Part C: Radiolabeling with ⁶⁴Cu for PET
- ⁶⁴Cu Chelation: Adjust the pH of ⁶⁴CuCl₂ solution to ~5.5 using ammonium acetate buffer. Add to the vancomycin-conjugated liposomes. Incubate at 40°C for 1 hour with shaking.
- Purification & Sterilization: Pass the reaction mixture through a PD-10 column (HBS eluent) to isolate radiolabeled liposomes from free ⁶⁴Cu. Filter sterilize using a 0.22 µm PES membrane.
- Quality Control:
- Size/PDI: Dynamic Light Scattering (DLS): Target ~110 nm, PDI < 0.15.
- Zeta Potential: Measure in HBS.
- Radiochemical Purity: Analyze via radio-instant thin-layer chromatography (radio-iTLC) using 50 mM EDTA as mobile phase. Purity must be >95%.
- Fluorescence Verification: Confirm ICG encapsulation via absorbance/emission spectra.
III. The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for Targeted Liposome Synthesis
| Item | Function & Critical Notes |
|---|---|
| DSPC (1,2-distearoyl-sn-glycero-3-phosphocholine) | Primary phospholipid forming the liposome bilayer; provides high phase transition temperature for stability. |
| DSPE-PEG2000-Maleimide | Functionalized PEG-lipid; provides steric stabilization (prevents opsonization) and presents maleimide group for thiol-based ligand conjugation. |
| Ammonium Sulfate ((NH₄)₂SO₄) Buffer | Used for remote (active) loading. Creates a pH gradient across the liposome membrane to trap ICG inside. |
| Traut's Reagent (2-Iminothiolane) | Thiolation reagent. Introduces a free sulfhydryl (-SH) group onto the vancomycin molecule for site-specific conjugation. |
| p-SCN-Bn-DOTA | Bifunctional chelator. The isothiocyanate (SCN) group couples to amine groups on the liposome surface, while the DOTA cage chelates ⁶⁴Cu. |
| PD-10 Desalting Columns | Size-exclusion chromatography columns for rapid buffer exchange and removal of small-molecule impurities (free dye, chelator, ligand). |
| Polycarbonate Extrusion Membranes (50-100 nm) | Porous membranes used with an extruder to produce liposomes with a uniform, defined size distribution, critical for consistent in vivo behavior. |
Visualized Workflows & Pathways
Title: Synthesis of Dual-Modal Vancomycin-Liposome Probe
Title: In Vivo Targeting & Multimodal Imaging Workflow
Within the broader thesis on bacteria-specific molecular imaging probes, translational research bridges the gap between bench-side discovery and clinical application. This document outlines the critical application notes and protocols for utilizing preclinical models and designing early-phase clinical trials to validate novel imaging probes for infection localization.
Application Notes: Preclinical Validation Workflow
Rationale for Model Selection
Preclinical models must recapitulate key aspects of human infection to provide predictive data for clinical trials. Selection depends on the target bacteria, infection site (e.g., soft tissue, biofilm on implant, pneumonia), and the probe's mechanism of action (e.g., targeting metabolic pathways, surface antigens).
Table 1: Common Preclinical Models for Infection Imaging Probe Validation
| Model Type | Infection Example | Key Advantages | Primary Limitations | Typical Readout Metrics |
|---|---|---|---|---|
| Murine Thigh Infection | S. aureus, P. aeruginosa | High-throughput, controllable inoculum. | Differences in host immunity vs. humans. | Target-to-Background Ratio (TBR), Biodistribution (%ID/g). |
| Rat/Murine Pneumonia | K. pneumoniae, S. pneumoniae | Models a complex, deep-seated infection. | Technical challenge for imaging. | Lung-specific signal, Correlation with bacterial CFU. |
| Rabbit Osteomyelitis/Implant | Methicillin-resistant S. aureus (MRSA) biofilm | Superior bone size for imaging, established biofilm models. | Higher cost, limited transgenic options. | Signal specificity vs. sterile inflammation, biofilm penetration. |
| Porcine Soft Tissue | Polymicrobial abscess | Similar skin physiology and thickness to humans. | Very high cost and resource-intensive. | Probe pharmacokinetics, spatial resolution of infection margins. |
Key Validation Parameters
Quantitative validation of imaging probes requires multi-modal assessment:
- Specificity: Signal in infected vs. sterile inflamed tissue.
- Sensitivity: Limit of detection (LoD) in terms of bacterial colony-forming units (CFU).
- Pharmacokinetics: Time-to-maximum uptake, clearance from blood and non-target tissues.
- Dosimetry: Radiation burden (for radiolabeled probes) or potential phototoxicity (for optical probes).
Table 2: Quantitative Benchmarks for Successful Preclinical Probe Validation
| Parameter | Optimal Target (Small Animal) | Measurement Technique | Justification |
|---|---|---|---|
| Target-to-Background Ratio (TBR) | > 3.0 (by 24h post-injection) | Quantitative region-of-interest (ROI) analysis on PET/SPECT/CT or fluorescence imaging. | Ensures sufficient contrast for reliable image interpretation. |
| Infected vs. Inflamed Tissue Signal Ratio | > 2.0 | Ex vivo gamma counting or fluorescence reflectance imaging of excised tissues. | Demonstrates bacteria-specific binding over nonspecific enhanced permeability and retention (EPR). |
| Correlation with Bacterial Burden (R²) | > 0.8 | Linear regression of imaging signal (e.g., %ID/g) vs. ex vivo CFU counts. | Validates that probe signal quantitatively reflects infection severity. |
| Blood Clearance Half-life (t1/2, β) | < 60 minutes (for radiolabeled probes) | Serial blood sampling and gamma counting or kinetic PET modeling. | Rapid clearance reduces background signal and improves TBR. |
Detailed Experimental Protocols
Protocol: Murine Thigh Infection Model for Probe Specificity Testing
Objective: To evaluate the specificity and pharmacokinetics of a radiolabeled bacteria-specific probe ([99mTc]Tc- or [68Ga]Ga-labeled) in a localized S. aureus infection.
Materials:
- Animals: Female BALB/c mice (6-8 weeks old).
- Bacteria: Staphylococcus aureus (e.g., strain ATCC 25923), prepared in mid-log phase.
- Probe: Radiolabeled candidate probe (e.g., 1-2 MBq/mouse in 100 µL saline).
- Control: Radiolabeled scrambled-sequence or non-targeting control probe.
- Imaging System: microSPECT/CT or microPET/CT scanner.
Procedure:
- Infection Induction: Anesthetize mouse. Inject 50 µL of S. aureus suspension (1x10^7 CFU) into the left posterior thigh muscle. For a sterile inflammation control, inject 50 µL of 3% aqueous thioglycollate into the right posterior thigh muscle of the same or a cohort animal.
- Probe Administration: At 24h post-infection (acute model), inject the radiolabeled probe via the tail vein.
- In Vivo Imaging: Acquire whole-body SPECT/PET scans at multiple time points (e.g., 1h, 4h, 24h post-injection). Perform a co-registered CT scan for anatomical localization.
- Ex Vivo Biodistribution: Euthanize animals at terminal time points (e.g., 4h and 24h). Excise infected thigh, inflamed thigh, major organs (blood, heart, lung, liver, spleen, kidney, muscle, bone). Weigh tissues and measure radioactivity using a gamma counter. Calculate percentage of injected dose per gram of tissue (%ID/g).
- CFU Determination: Homogenize the excised infected thigh tissue in PBS, plate serial dilutions on agar plates, and incubate overnight for CFU counting.
Protocol: Design of a First-in-Human (FIH) Phase I Trial for an Imaging Probe
Objective: To assess the safety, pharmacokinetics, and preliminary imaging efficacy of a novel [18F]-labeled bacteria-specific tracer in healthy volunteers and patients with suspected orthopedic infections.
Study Design: Open-label, non-randomized, sequential cohort study. Cohorts:
- Cohort A (n=8): Healthy volunteers. Single ascending doses of the probe.
- Cohort B (n=10): Patients with suspected prosthetic joint infection (PJI) scheduled for revision surgery.
Key Procedures:
- Dose Administration: Intravenous bolus injection of the probe. Dose levels based on preclinical dosimetry (e.g., starting at 1/50th of the no-observed-adverse-effect-level dose from rodent studies).
- Safety Monitoring: Continuous monitoring for 2h post-injection (vitals, ECG, blood work at baseline, 1h, 24h). Record all adverse events for 7 days.
- Imaging Protocol: Whole-body PET/MRI or PET/CT scans at 0-60 min (dynamic) and 120 min post-injection. For patients, a focused scan on the affected joint at 120 min is added.
- Pharmacokinetic Analysis: Serial venous blood sampling over 3h. Generate time-activity curves for blood and major organs. Calculate standard PK parameters (AUC, Cmax, t1/2).
- Image Analysis: Qualitative assessment of image quality and abnormal uptake. Quantitative analysis (SUVmax, SUVpeak, TBR) of suspected infection sites in patients.
- Correlative Microbiology: For Cohort B, imaging findings are correlated with intraoperative tissue culture and histopathology as the diagnostic standard.
Visualization Diagrams
Title: Translational Pathway for Imaging Probes
Title: Phase I Clinical Trial Workflow for Infection Probe
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Preclinical and Early-Clinical Translation of Infection Imaging Probes
| Item / Reagent | Function & Application | Key Considerations |
|---|---|---|
| Cyclotron & Radiochemistry Module | Production of short-lived radionuclides (e.g., 18F, 68Ga) and radiosynthesis of the probe. | GMP-compliance is critical for clinical trial material production. |
| Good Manufacturing Practice (GMP) Facilities | For synthesis, purification, and quality control of the imaging probe for human use. | Ensures product safety, identity, strength, purity, and quality. |
| Animal Imaging Systems (microPET/SPECT/CT, FMT) | Non-invasive, longitudinal quantification of probe biodistribution and kinetics in preclinical models. | Multi-modal systems (PET/CT, PET/MRI) provide superior anatomical context. |
| Clinical PET/CT or PET/MRI Scanner | High-resolution imaging of probe distribution in human patients. | Required for Phase I/II trials to obtain diagnostic-quality images. |
| Validated Bacterial Strains (ATCC) | Well-characterized strains for consistent infection model induction (e.g., S. aureus USA300, P. aeruginosa PAO1). | Use of clinically relevant, including antibiotic-resistant, strains is essential. |
| Specialized Animal Diet (e.g., Alfalfa-Free) | Minimizes autofluorescence in optical imaging studies, reducing background signal. | Critical for achieving high sensitivity in fluorescence-based imaging protocols. |
| Automated Gamma Counter & HPLC | For ex vivo biodistribution analysis and in vitro radiochemical purity/p stability testing. | High-throughput counters improve efficiency for multi-time-point studies. |
| Electronic Data Capture (EDC) System | For secure and compliant collection of clinical trial data (safety, PK, imaging reads). | Must be 21 CFR Part 11 compliant for regulatory submission. |
Navigating Challenges: Optimization Strategies for Specificity and Sensitivity
Within the development of bacteria-specific molecular imaging probes for infection localization, a paramount challenge is the high degree of non-specific binding (NSB) to host tissues and biomolecules. This background signal obscures the specific detection of pathogenic bacteria, reducing the signal-to-noise ratio (SNR) and diagnostic accuracy. This application note details validated strategies and protocols to overcome host background, focusing on surface passivation, probe engineering, and blocking techniques critical for in vivo and ex vivo imaging research.
Quantitative Analysis of Common Strategies
The efficacy of various NSB reduction strategies is quantified below, based on current literature and experimental data.
Table 1: Efficacy of Non-Specific Binding Reduction Strategies
| Strategy | Mechanism | Typical Reduction in NSB (%) | Resultant SNR Improvement (Fold) | Key Applications |
|---|---|---|---|---|
| Polyethylene Glycol (PEG)ylation | Hydrophilic polymer shield sterically repels proteins. | 60-85% | 3-8x | In vivo probe circulation, surface coating of nanoparticles. |
| Protein-Based Blocking (BSA, Casein) | Saturates non-specific sites on surfaces and tissues. | 40-70% | 2-5x | Ex vivo tissue staining, immunoassays, blotting. |
| Charge Optimization (Zwitterionic Ligands) | Presents neutral net charge, minimizing electrostatic interactions. | 70-90% | 5-10x | Quantum dots, fluorescent dye conjugation, implant coatings. |
| Affinity Purification (e.g., via His-Tag) | Removes unconjugated, reactive dye molecules. | 30-50% | 1.5-3x | All probe conjugation protocols. |
| Pre-injection of Clearing Agents | Binds and neutralizes circulating host factors (e.g., serum proteins). | 50-75% | 2-6x | In vivo imaging, particularly in reticuloendothelial system-rich areas. |
| Use of *F(ab')2 Fragments* | Removes Fc region responsible for non-specific Fc receptor binding. | 50-80% | 2-7x | Antibody-based probes for infection imaging. |
Detailed Experimental Protocols
Protocol 3.1: Zwitterionic Surface Modification of Iron Oxide Nanoparticles forIn VivoImaging
Objective: To coat nanoparticle probes with zwitterionic polymers to minimize serum protein adsorption (opsonization) and reduce uptake by host macrophages.
Materials:
- Carboxyl-terminated iron oxide nanoparticles (10 nm, 1 mg/mL in MES buffer).
- Zwitterionic polymer (e.g., Poly(carboxybetaine methacrylate), PCB, 10 kDa).
- 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and N-Hydroxysuccinimide (NHS).
- Phosphate Buffered Saline (PBS), pH 7.4.
- Zeba Spin Desalting Columns (7K MWCO).
Procedure:
- Activation: Mix 1 mL of nanoparticle solution with 5 mM EDC and 2 mM NHS. React for 15 minutes at room temperature (RT) with gentle shaking.
- Conjugation: Add PCB polymer at a 50:1 molar excess (polymer to estimated nanoparticle number). Incubate for 2 hours at RT.
- Purification: Pass the reaction mixture through a Zeba column pre-equilibrated with PBS to remove excess reactants and byproducts. Centrifuge at 1500 x g for 2 minutes.
- Characterization: Measure hydrodynamic diameter and zeta potential via Dynamic Light Scattering (DLS). Successful coating is indicated by a near-neutral zeta potential (±5 mV) and reduced protein adsorption in 100% FBS after 1-hour incubation compared to uncoated controls.
- Validation: In a murine myositis model, inject modified nanoparticles conjugated to a vancomycin derivative. Image via MRI. SNR in infected muscle should be ≥5x higher than for uncoated probes in contralateral tissue.
Protocol 3.2: Multi-Step Blocking forEx VivoFluorescent Probe Staining of Bacterial Biofilms in Tissue
Objective: To minimize non-specific binding of fluorescently-labeled antimicrobial peptides (AMPs) to mammalian tissue sections for clear biofilm visualization.
Materials:
- Fresh-frozen tissue sections (e.g., murine catheter-associated infection model).
- Fluorescently-labeled AMP probe (e.g., labeled with CF488A).
- Blocking Buffer: 3% (w/v) Bovine Serum Albumin (BSA) + 1% (w/v) Casein in Tris-Buffered Saline with 0.05% Tween-20 (TBST).
- Pre-absorption Serum: 2% normal serum from the host species (e.g., mouse serum) in TBST.
- Mounting medium with DAPI.
Procedure:
- Fixation & Permeabilization: Fix tissue sections in 4% PFA for 15 min, then permeabilize with 0.1% Triton X-100 for 10 min. Wash 3x with PBS.
- Pre-Absorption Block: Incubate sections with pre-absorption serum for 1 hour at RT in a humidified chamber. This blocks species-specific reactive sites.
- Primary Block: Rinse briefly with TBST. Incubate with the protein-based Blocking Buffer for 2 hours at RT.
- Probe Incubation: Dilute the fluorescent AMP probe in fresh Blocking Buffer. Apply to tissue section and incubate overnight at 4°C in the dark.
- Washing: Wash stringently 5x with TBST, 5 minutes per wash, with gentle agitation.
- Imaging: Mount with DAPI-containing medium. Image using a confocal microscope. Specific bacterial signal (e.g., green) should be distinctly localized from host tissue autofluorescence, with minimal probe adhesion to mammalian cells.
Visualization of Strategies and Workflows
Diagram Title: NSB Reduction Strategy Decision Workflow
Diagram Title: Mechanism of Zwitterionic Coating for Stealth
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Reducing Non-Specific Binding
| Reagent / Material | Primary Function | Application Context |
|---|---|---|
| mPEG-NHS Ester (e.g., 5kDa) | Conjugates to amine groups on probes/proteins, creating a hydrophilic, steric barrier. | PEGylation of antibodies, peptides, and nanoparticle surfaces. |
| Zwitterionic Polymer (e.g., PCB-NHS) | Provides a super-hydrophilic, charge-balanced coating that resists protein adsorption. | Creating "stealth" coatings for in vivo imaging probes and implants. |
| Bovine Serum Albumin (BSA), Fraction V | Inexpensive, general-purpose blocking protein that adsorbs to hydrophobic surfaces. | Blocking in Western blotting, ELISA, and ex vivo tissue staining. |
| Casein (from Milk) | Effective blocker for charged nylon/PVDF membranes and tissues; reduces electrostatic NSB. | Alternative or supplement to BSA, especially for phospho-specific antibodies. |
| Normal Serum (Host Species Matched) | Contains immunoglobulins that pre-absorb anti-species reactive sites in tissues. | Pre-blocking step in immunohistochemistry/immunofluorescence. |
| Zeba Spin Desalting Columns | Rapid buffer exchange and removal of small molecule contaminants (dyes, crosslinkers). | Purifying conjugated probes after labeling reactions. |
| Tween-20 or Triton X-100 | Non-ionic detergents that reduce hydrophobic interactions and permeabilize membranes. | Component of washing and blocking buffers (e.g., TBST, PBST). |
| HisTrap HP Column | Affinity chromatography resin for purifying His-tagged proteins from reaction mixtures. | Removing unconjugated fluorescent dyes or drugs from labeled protein probes. |
Within the development of bacteria-specific molecular imaging probes for infection localization, the central challenge is achieving an optimal signal-to-background ratio (SBR). This is dictated by the pharmacokinetic (PK) balance: rapid blood clearance of unbound probe to reduce background signal versus sufficient retention at the bacterial target site to generate a detectable signal. This Application Note details strategies and protocols for fine-tuning these PK parameters, focusing on chemical modifications, linker systems, and validation methods critical for creating effective infection imaging agents.
Core Strategies for PK Fine-Tuning
Chemical Modifications to Modulate Blood Clearance
| Strategy | Mechanism of Action | Effect on Blood Clearance | Effect on Target Retention | Key Trade-offs |
|---|---|---|---|---|
| PEGylation | Increases hydrodynamic radius, reduces renal filtration and opsonization. | Decreases (slower) | Often decreases (steric hindrance of binding) | Reduced immunogenicity vs. potentially reduced affinity. |
| Albumin Binding Motifs | Creates reversible binding to serum albumin. | Decreases (slower, prolonged circulation) | Can increase via enhanced permeability and retention (EPR) at infected sites. | Risk of high background signal; requires careful affinity tuning. |
| Charge Modulation | Adjusting net charge (e.g., cationic for bacterial membrane interaction). | Variable; cationic often increases clearance via RES uptake. | Increases (electrostatic binding to anionic bacterial surfaces). | Risk of non-specific binding to host tissues. |
| Hydrophilicity/Lipophilicity | Adjusting LogP via functional groups. | Increased hydrophilicity favors renal clearance; lipophilicity favors hepatobiliary clearance. | Lipophilicity may increase non-specific tissue retention. | Must balance for desired clearance route (renal vs. hepatic). |
| Probe Size Tuning | Using small molecules, peptides, or antibody fragments. | Small molecules/peptides: Fast clearance. Antibody fragments: Intermediate. Full antibodies: Slow. | Affinity-driven; smaller probes may have faster washout from target. | Smaller probes enable faster imaging protocols. |
Targeting Moieties and Linker Systems
| Targeting Moety | Example Target (Bacteria) | Typical PK Profile | Optimal Use Case |
|---|---|---|---|
| Small Molecules | S. aureus-targeted trimethoprim, Folate analogs for certain species. | Rapid blood clearance (minutes-hours). | Acute infection imaging, rapid-turnaround protocols. |
| Peptides | Antimicrobial peptides (e.g., UBI 29-41), Peptide nucleic acids. | Fast to intermediate clearance (minutes-1 hour). | Flexible, can be engineered for specific clearance rates. |
| Antibody Fragments (Fab, scFv, VHH) | Anti-S. aureus VHH, Anti-lipoteichoic acid Fab. | Intermediate clearance (1-6 hours). | High specificity with faster kinetics than full antibodies. |
| Full Antibodies | Anti-staphylococcal monoclonal antibodies. | Very slow clearance (days). | Generally suboptimal for imaging due to high background. |
Linker Chemistry: The linker between targeting moiety and reporter (e.g., fluorophore, radionuclide chelator) is critical. Cleavable linkers (e.g., enzyme-responsive) can be used to trap the reporter at the target site, enhancing retention after blood clearance.
Quantitative PK Parameters for Probe Evaluation
Table 1: Key In Vivo Pharmacokinetic Parameters for Probe Assessment
| Parameter | Symbol | Definition | Desired Profile for Infection Imaging | Typical Measurement Method |
|---|---|---|---|---|
| Area Under Curve | AUC | Total exposure over time. | Minimized for blood, maximized for target tissue ratio. | Blood sampling, imaging-derived Time-Activity Curves (TACs). |
| Half-Life | t1/2,α, t1/2,β | Distribution/elimination half-life. | Short distribution, appropriate elimination to match imaging window. | Non-compartmental analysis of blood TACs. |
| Clearance | CL | Volume of blood cleared per unit time. | High total clearance. | Dose / AUC. |
| Volume of Distribution | Vd | Apparent volume into which probe distributes. | Sufficient to access extravascular infection sites. | Dose / (AUC * kel). |
| Target-to-Background Ratio | TBR | (Signal in Target Tissue) / (Signal in Background Tissue). | Maximized at optimal imaging time point. | Ex vivo biodistribution or quantitative imaging (e.g., PET/CT). |
| % Injected Dose per Gram | %ID/g | Uptake in specific tissue. | High in target infection, low in blood and non-target organs. | Ex vivo gamma counting or fluorescence spectroscopy. |
Experimental Protocols
Protocol 4.1: In Vivo Pharmacokinetic and Biodistribution Study in a Murine Myositis Model
Objective: To determine blood clearance kinetics and tissue distribution profile of a candidate bacteria-specific imaging probe.
Materials:
- Animal Model: Mice with unilateral thigh bacterial infection (e.g., S. aureus) and contralateral sterile inflammation control.
- Candidate Probe: Radiolabeled (e.g., ⁹⁹ᵐTc, ⁶⁸Ga, ¹¹¹In) or fluorescently labeled bacteria-targeting agent.
- Control Probe: Isotype control or scrambled sequence probe.
- Instruments: Gamma counter, HPLC system, microcentrifuge, IV injection setup.
Procedure:
- Probe Administration: Inject ~100 µL of probe solution (1-100 pmol, 10-100 µCi) via tail vein.
- Blood Sampling: Collect blood samples (10-20 µL) retro-orbitally or from tail nick at 1, 5, 15, 30, 60, 120, and 240 minutes post-injection (p.i.). Immediately dilute in pre-weighed tubes with PBS.
- Processing Blood: Weigh tubes, measure radioactivity/fluorescence. Express data as %ID/g.
- Terminal Biodistribution: At predetermined time points (e.g., 1h, 4h, 24h p.i.), euthanize animals (n=5/group). Excise tissues of interest: blood, heart, lungs, liver, spleen, kidneys, muscle (infected and control), bone.
- Tissue Analysis: Weigh tissues, measure radioactivity/fluorescence. Calculate %ID/g and TBR (Infected Muscle/Control Muscle; Infected Muscle/Blood).
- Metabolite Analysis (Optional): At early time points, collect blood and urine, analyze via radio-HPLC to determine probe stability in vivo.
Protocol 4.2: Ex Vivo Binding Affinity and Specificity Assay
Objective: To correlate in vivo retention with in vitro affinity and specificity for bacterial cells.
Materials:
- Bacterial strains (target and non-target controls).
- Cultured mammalian cells (e.g., macrophages, myocytes).
- Labeled probe.
- Flow cytometer or microplate reader.
Procedure:
- Bacterial Culture: Grow bacteria to mid-log phase, wash, and resuspend in binding buffer.
- Saturation Binding: Incubate serial dilutions of labeled probe with a fixed number of bacteria (10⁸ CFU) for 1h at 37°C.
- Washing: Pellet bacteria, wash 2x with buffer to remove unbound probe.
- Measurement: Quantify cell-associated signal. Use non-specific binding controls (excess unlabeled competitor).
- Data Analysis: Perform nonlinear regression to determine equilibrium dissociation constant (KD) and Bmax.
- Specificity: Repeat with non-target bacterial strains and mammalian cells to assess off-target binding.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for PK Tuning Studies
| Item | Function in PK Studies | Example Product/Category |
|---|---|---|
| Site-Specific Bioconjugation Kits | Enables controlled attachment of PK-modifying groups (PEG, albumin binders) or labels to targeting moieties without impairing function. | Click chemistry kits, enzyme-based ligation kits (Sortase, Transglutaminase). |
| Modular Linker Reagents | Provides chemical spacers and cleavable linkers (e.g., cathepsin-sensitive, pH-sensitive) to engineer probe stability and retention. | PEG linkers, Val-Cit-PABC linker, hydrazone linkers. |
| Radionuclide Chelators | For radiometal labeling (⁶⁸Ga, ⁶⁴Cu, ¹¹¹In) enabling sensitive quantitative PK tracking. | DOTA, NOTA, DFO chelators. |
| Near-Infrared Fluorescent Dyes | For optical imaging studies; Cy7, IRDye 800CW derivatives allow deep tissue signal detection. | Sulfo-Cy7 NHS ester, IRDye 800CW Maleimide. |
| Albumin from Mouse/Serum | Used in in vitro assays to test the impact of albumin binding on probe function and stability. | Fatty-acid free BSA or species-specific serum. |
| Protease/Plasma Stability Assay Kits | To assess probe stability in blood serum, a key determinant of circulation half-life. | Human/mouse plasma stability assay kits. |
| In Vivo Imaging Systems | For non-invasive, longitudinal PK and biodistribution studies (optical, PET, SPECT). | IVIS Spectrum, microPET/CT, microSPECT/CT. |
Visualizations
Diagram Title: PK Fine-Tuning Optimization Workflow
Diagram Title: Modular Probe Design Dictates PK Metrics
Application Notes
Within the research paradigm of developing bacteria-specific molecular imaging probes for infection localization, a central challenge is bacterial heterogeneity. This refers to the vast diversity in cell wall structure, metabolic activity, genetic expression, and enzymatic profiles across different bacterial species and even within populations of the same species. This heterogeneity directly informs the strategic choice between designing broad-spectrum probes (targeting conserved microbial features) and narrow-spectrum probes (targeting pathogen-specific markers).
Broad-Spectrum Probe Strategy: This approach aims for a "pan-bacterial" imaging agent capable of detecting a wide range of Gram-positive and Gram-negative pathogens. The primary advantage is utility in first-line diagnostic imaging where the causative agent is unknown. Key targets include:
- Conserved Structural Components: e.g., peptidoglycan (targeted by vancomycin derivatives, D-amino acids), lipopolysaccharides (LPS) in Gram-negatives (targeted by polymyxin derivatives).
- Core Metabolic Pathways: e.g., bacterial folate synthesis (targeted by trimethoprim analogs), or prokaryotic protein synthesis machinery.
- Universal Enzymatic Activity: e.g., bacterial-specific phosphatases, β-lactamases, or reductase enzymes that can activate a probe.
The risk lies in potential off-target binding to host cells or commensal flora, and missing atypical or resistant strains lacking the targeted universal feature.
Narrow-Spectrum Probe Strategy: This approach designs probes for precise pathogen identification, crucial for targeted therapy and antimicrobial stewardship. It aligns with the precision medicine ethos. Key targets include:
- Species-Specific Surface Antigens: e.g., Staphylococcus aureus-specific antibodies or affinity molecules.
- Pathogen-Specific Virulence Factors: e.g., toxins or secretion systems unique to a pathogen.
- Strain-Specific Enzymatic Signatures: e.g., enzymes associated with specific antibiotic resistance profiles (e.g., Klebsiella pneumoniae carbapenemase).
The primary challenge is the need for a priori clinical suspicion of the pathogen and the resource-intensive development of multiple probes for clinical use.
Quantitative Comparison of Probe Design Strategies: Table 1: Comparative Analysis of Broad vs. Narrow-Spectrum Imaging Probe Strategies
| Parameter | Broad-Spectrum Probes | Narrow-Spectrum Probes |
|---|---|---|
| Primary Target Examples | Peptidoglycan, LPS, bacterial reductase | Species-specific antigens, virulence factors |
| Theoretical Coverage | High (60-90% of common pathogens) | Low to Moderate (Single species or strain) |
| Clinical Use Case | First-line infection localization, unknown pathogen | Pathogen identification, therapy guidance |
| Development Complexity | Moderate | High (Requires specific ligand discovery) |
| Risk of Off-Target Binding | Higher (e.g., host mitochondria, commensals) | Lower (if target is truly unique) |
| Susceptibility to Resistance | Lower (targets essential structures) | Higher (targets can be downregulated) |
| Exemplary Probe (Research) | [¹⁸F]FDG-labeled vancomycin | Anti-S. aureus antibody-⁶⁸Ga conjugate |
| Key Performance Metric (in vivo) | Sensitivity >85%, Specificity ~70-80% | Sensitivity >95%, Specificity >90% |
Table 2: Performance Metrics of Selected Probe Candidates from Recent Literature (2022-2024)
| Probe Name / Core | Spectrum | Target / Mechanism | Model (In Vivo) | Signal-to-Background Ratio (Mean ± SD) | Reference (Type) |
|---|---|---|---|---|---|
| [⁶⁸Ga]Ga-DOTA-Ubiquicidin (29-41) | Broad | Disrupts bacterial membrane | S. aureus thigh infection (Mouse) | 3.5 ± 0.8 | Clinical Trial Phase II |
| [¹⁸F]Fluoromaltotriose | Narrow | E. coli maltodextrin transporter | E. coli pyelonephritis (Rat) | 4.2 ± 1.1 | Research Article |
| ⁹⁹ᵐTc-labeled Ciprofloxacin | Broad | Binds to DNA gyrase in live bacteria | P. aeruginosa lung infection (Mouse) | 2.8 ± 0.5 | Research Article |
| IR800-WCA | Broad | Binds to Gram-positive peptidoglycan | S. aureus vs. E. coli (Mouse) | 5.1 ± 1.3 (Gram+) | Research Article |
| sulfo-Cy5-PV | Narrow | Activates Vibrio cholerae’s S.T. effector | V. cholerae colonization (Mouse) | >10.0 | Research Article |
Experimental Protocols
Protocol 1: In Vitro Validation of Broad-Spectrum Probe Specificity Using a Panel of Bacterial Strains
Objective: To assess the binding affinity and specificity of a candidate broad-spectrum probe (e.g., a fluorescent vancomycin derivative) against a heterogeneous panel of bacteria.
Materials: See The Scientist's Toolkit below. Procedure:
- Bacterial Culture: Inoculate 5 mL of appropriate broth (e.g., TSB, LB) with a single colony of each target strain (S. aureus ATCC 29213, E. coli ATCC 25922, P. aeruginosa PAO1, E. faecalis ATCC 29212). Incubate at 37°C with shaking (200 rpm) to mid-log phase (OD₆₀₀ ≈ 0.5-0.6).
- Cell Harvesting & Washing: Pellet 1 mL of each culture by centrifugation (5,000 x g, 5 min, 4°C). Gently wash the bacterial pellet twice with 1x PBS (pH 7.4). Resuspend the final pellet in 1 mL of PBS and adjust the cell density to ~1 x 10⁸ CFU/mL using McFarland standards or OD₆₀₀ correlation.
- Probe Incubation: Aliquot 100 µL of each bacterial suspension into a microcentrifuge tube. Add the fluorescent probe (e.g., BODIPY-FL-vancomycin) to a final concentration of 1 µM. Include control tubes: bacteria only (no probe) and probe only (no bacteria). Incubate in the dark at 37°C for 30 minutes.
- Washing & Signal Measurement: Pellet cells (10,000 x g, 3 min). Wash twice with 500 µL PBS to remove unbound probe. Finally, resuspend in 200 µL PBS. Transfer 100 µL of each suspension to a black-walled, clear-bottom 96-well plate in triplicate.
- Quantification: Measure fluorescence intensity using a plate reader (λₑₓ/λₑₘ per fluorophore, e.g., 485/520 nm for BODIPY-FL). Calculate the mean fluorescence intensity (MFI) for each strain after subtracting the background (bacteria-only control).
- Confocal Microscopy Validation: For qualitative imaging, perform a parallel incubation, wash, and mount 5 µL of the final suspension on a glass slide with a coverslip. Image using a confocal microscope with appropriate laser and filter settings.
Protocol 2: In Vivo Optical Imaging of a Targeted Infection Using a Narrow-Spectrum Activatable Probe
Objective: To visualize a specific bacterial infection (e.g., E. coli) in a live mouse model using a narrow-spectrum, enzyme-activatable probe (e.g., fluorogenic substrate for β-glucuronidase).
Materials: See The Scientist's Toolkit below. Procedure:
- Infection Model Establishment: Anesthetize an 8-week-old female BALB/c mouse. For a thigh infection model, inject 50 µL of a suspension containing ~1 x 10⁷ CFU of a β-glucuronidase-positive E. coli strain into the left posterior thigh muscle. Inject 50 µL of sterile PBS into the right thigh as a control. Allow infection to establish for 6-8 hours.
- Probe Administration: Prepare a solution of the activatable probe (e.g., Cy5.5-β-D-glucuronide) in sterile saline. Inject the probe via the tail vein at a dose of 2 nmol (in 100 µL volume).
- In Vivo Optical Imaging: Anesthetize the mouse at designated time points post-injection (e.g., 1, 4, 24 h). Place the mouse in the imaging chamber of a pre-warmed (37°C) IVIS Spectrum or similar system. Acquire fluorescence images using appropriate filters (λₑₓ = 675 nm, λₑₘ = 720 nm for Cy5.5). Acquire a white-light photograph for overlay. Maintain anesthesia with 1-2% isoflurane.
- Ex Vivo Validation: After the final imaging time point, euthanize the mouse humanely. Excise the infected and control thigh muscles, as well as major organs (liver, spleen, kidneys). Rinse in PBS, blot dry, and place in the imaging system to quantify ex vivo fluorescence signal. Homogenize tissues to determine bacterial CFU/g to correlate signal with bacterial burden.
- Data Analysis: Use imaging software to draw regions of interest (ROIs) over the infection and control sites. Quantify signal as total radiant efficiency ([p/s/cm²/sr] / [µW/cm²]). Express data as target-to-background ratio (TBR): Signal(Infected Thigh) / Signal(Contralateral Thigh).
Diagrams
Diagram 1: Probe Design Strategy Decision Flowchart
Diagram 2: Mechanism of a Narrow-Spectrum Activatable Probe
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for Bacterial Probe Development
| Item | Function / Application | Example Product / Specification |
|---|---|---|
| Fluorescent Antibiotic Conjugates | Serve as scaffold for broad-spectrum probe development; bind conserved targets. | BODIPY-FL Vancomycin, NBD-labeled Polymyxin B. |
| Activatable (Smart) Probes | Enable high SBR imaging via specific enzymatic activation by pathogen. | β-lactamase (e.g., BlaC) or β-glucuronidase cleavable NIR fluorogenic substrates. |
| D-Amino Acid Derivatives (DAAs) | Incorporate into bacterial peptidoglycan; backbone for broad-spectrum probes. | HADA (7-hydroxycoumarin-3-carboxylic acid-D-lysine), NBD-amino-D-alanine. |
| Siderophore-Based Chelators | Exploit iron acquisition systems for targeted delivery, esp. in Gram-negatives. | Deferoxamine (DFO) conjugates for ⁶⁸Ga labeling; Enterobactin analogs. |
| Near-Infrared (NIR) Fluorophores | Minimize tissue autofluorescence for deep-tissue optical imaging in vivo. | Cy5.5, Cy7, IRDye 800CW, CF dyes (λₑₘ > 650 nm). |
| Radionuclide Chelators | Enable conjugation of radioisotopes for PET/SPECT imaging. | DOTA, NOTA, DTPA for ⁶⁸Ga, ⁶⁴Cu; Isothiocyanate-benzyl-DFO. |
| Bioluminescent Bacterial Strains | Provide a reference gold standard for infection localization in animal models. | Xenogen-lux transformed S. aureus (Xen29) or E. coli (Xen14). |
| IVIS Spectrum Imaging System | In vivo and ex vivo quantitative 2D/3D optical imaging (fluorescence + bioluminescence). | PerkinElmer IVIS SpectrumCT. |
Addressing Biofilm Penetration and Hypoxic Microenvironments
Within the broader thesis on developing bacteria-specific molecular imaging probes for infection localization, a paramount challenge is the probe’s ability to target bacteria residing within structured biofilms and hypoxic niches. These environments, common in chronic infections like those associated with cystic fibrosis, orthopedic implants, and diabetic wounds, severely limit the efficacy of both diagnostics and therapeutics. This application note details protocols and strategies for evaluating and enhancing probe penetration and activation within these barriers.
Table 1: Characteristics of Biofilm and Hypoxic Microenvironments Impeding Probe Delivery
| Parameter | Typical Range in Mature Biofilms | Impact on Imaging Probes |
|---|---|---|
| Diffusion Barrier (EPS) | EPS thickness: 10 - 200 µm | Reduces probe diffusion coefficient by 10- to 100-fold compared to aqueous solution. |
| Hypoxic Zones | Oxygen concentration: <0.5% - 1.0% (vs. ~21% in air) | Renders oxygen-dependent fluorescence (e.g., from GFP-like proteins) ineffective; necessitates anaerobic-activatable probes. |
| Redox Potential | -200 mV to -400 mV (highly reducing) | Can prematurely activate or deactivate redox-sensitive probe linkers. |
| pH | 5.5 - 7.0 (can be acidic in deeper layers) | Can affect probe solubility, charge, and fluorescence quantum yield. |
| Bacterial Density | 10^8 - 10^11 CFU/mL in biofilm matrix | High background if probe has non-specific binding; requires high specificity for bacterial vs. host cells. |
Table 2: Strategies for Enhanced Biofilm Penetration
| Strategy | Mechanism | Example Probes/Agents |
|---|---|---|
| EPS Matrix Degradation | Use of enzymes (DNase, dispersin B) to degrade extracellular DNA or polysaccharides. | DNase I co-administration with fluorescent vancomycin. |
| Size Reduction | Use of small molecule probes or nanoparticle size optimization (<20 nm). | Nitroimidazole-based hypoxia probes (MW <500 Da). |
| Charge Modulation | Cationic probes to interact with negatively charged EPS, or zwitterionic coatings to reduce non-specific binding. | Zwitterionic-coated quantum dots conjugated with antimicrobial peptides. |
| Triggered Activation | Probes activated specifically by bacterial enzymes (e.g., β-lactamase, lipase) or low pH. | β-lactamase-activated fluorescent probes (e.g., CCF4 derivatives). |
Experimental Protocols
Protocol 1: Evaluating Probe Penetration in anIn VitroBiofilm Model
Objective: To quantitatively assess the depth and uniformity of probe distribution within a standard biofilm.
Materials:
- Bacterial strain (e.g., Pseudomonas aeruginosa PAO1, Staphylococcus aureus)
- Growth medium (e.g., Tryptic Soy Broth, LB)
- Flow cell or 96-well μ-plate for static biofilm
- Test imaging probe (fluorescent or radioactive label)
- Confocal Laser Scanning Microscope (CLSM)
- Image analysis software (e.g., ImageJ, Imaris)
Procedure:
- Biofilm Formation: Inoculate flow cells or well plates with mid-log phase bacteria. For flow cells, perfuse with medium for 24-72 hrs. For static biofilms, incubate with medium renewal every 24 hrs.
- Probe Administration: Dilute the probe in relevant buffer (e.g., PBS). Apply to the biofilm system. For static biofilms, replace medium with probe solution. For flow cells, perfuse at a low shear rate (e.g., 0.2 mL/min) for 1-4 hours.
- Washing: Gently perfuse or rinse with buffer to remove unbound probe.
- Imaging: Using CLSM, acquire Z-stacks (step size: 1-2 µm) of multiple random fields.
- Quantitative Analysis:
- Use software to plot fluorescence intensity as a function of depth from the biofilm surface to the substratum.
- Calculate the Penetration Efficiency (PE):
(Area under the intensity-depth curve for test probe) / (Area under the curve for a small, non-binding control dye, e.g., fluorescein) x 100%. - Generate 2D false-color maps of fluorescence distribution.
Protocol 2: Validating Probe Activation in Hypoxic Conditions
Objective: To confirm that a hypoxia-activatable probe (e.g., nitroreductase-sensitive) selectively signals under low oxygen.
Materials:
- Anaerobic chamber or sealed hypoxia incubator
- Bacterial cultures (aerobic and anaerobic strains)
- Hypoxia probe (e.g., Nitroreductase (NTR)-responsive probe like Cy7-NO₂)
- Normoxic control probe (non-activatable analog)
- Microplate reader or in vivo imaging system (IVIS)
Procedure:
- Sample Preparation: Prepare bacterial suspensions (~10⁸ CFU/mL) in sealed vials or 96-well plates.
- Oxygen Control: Place one set in a normoxic incubator (21% O₂). Place another set in an anaerobic chamber (<1% O₂) or hypoxia incubator (1-5% O₂) for 4-6 hours to induce bacterial adaptation and hypoxia.
- Probe Incubation: Add the hypoxia probe and control probe to separate wells/vials. Incubate under respective oxygen conditions for 1-2 hours.
- Signal Measurement: For in vitro plates, read fluorescence/ luminescence in a pre-equilibrated plate reader. For in vivo validation, image animals (if applicable) using IVIS.
- Data Analysis: Calculate the Hypoxic Activation Ratio:
(Signal from hypoxia probe in low O₂) / (Signal from hypoxia probe in normoxia). A ratio >5 is typically considered indicative of specific activation.
Visualization: Pathways and Workflows
Diagram 1: Hypoxia-Activatable Probe Mechanism
Diagram 2: Workflow for Probe Evaluation in Biofilms
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Biofilm/Hypoxia Probe Research
| Item | Function & Relevance | Example/Supplier |
|---|---|---|
| Calgary Biofilm Device | High-throughput assay for growing 96 identical biofilms for MIC/MBC and penetration testing. | Innovotech; MBEC Assay System. |
| Nitroreductase (NTR) Enzyme | Positive control for validating hypoxia-activatable probe mechanisms in vitro. | Recombinant E. coli NTR (Sigma-Aldrich). |
| Dispersin B (DspB) | Glycoside hydrolase that degrades poly-N-acetylglucosamine (PNAG), a key EPS component in many biofilms. | Used as a penetration enhancer; available from biotech suppliers. |
| Tetrazolium Salts (CTC, XTT) | Indicators of metabolic activity in biofilms; used to correlate probe localization with viable bacterial zones. | CTC (5-Cyano-2,3-ditolyl tetrazolium chloride) for microscopy. |
| Hypoxia-Inducible Factor (HIF-1α) Antibodies | Immunohistochemical validation of hypoxic regions in tissue infection models co-localized with probe signal. | Available from major antibody suppliers (CST, Abcam). |
| Synthetic Cystic Fibrosis Sputum Medium | Physiologically relevant culture medium for growing P. aeruginosa biofilms mimicking in vivo conditions. | Formulations available in literature (e.g., SCFM2). |
| Matrigel / Collagen Embedding | For creating 3D ex vivo models that mimic tissue density and diffusion barriers. | Corning Matrigel. |
| Oxygen-Sensitive Nanoparticles | Reference standards for mapping oxygen gradients in biofilms (e.g., via phosphorescence lifetime imaging). | NanO2-IR (Oxygen Enterprises). |
Within the broader research on bacteria-specific molecular imaging probes for infection localization, a critical translational gap exists between promising in vitro radiotracers and those suitable for human clinical trials. This document details the application notes and protocols required to navigate the complex pathway from laboratory-scale synthesis to the production of clinical-grade tracers under Good Manufacturing Practice (GMP). The focus is on scalability, quality control, and regulatory compliance, specifically for novel probes like radiolabeled maltose derivatives, trimethoprim analogs, or parmaceutical-specific agents designed to differentiate bacterial infection from sterile inflammation.
Key Challenges in Scale-Up and GMP Translation
The transition from lab synthesis to GMP production introduces multi-faceted challenges. The table below summarizes the primary constraints and requirements.
Table 1: Key Challenges in Scaling from Research to GMP-Grade Tracer Production
| Aspect | Lab-Scale (Research) | GMP Clinical Production | Primary Scaling Challenge |
|---|---|---|---|
| Synthesis Environment | Open system, fume hood. | ISO-classified cleanrooms (ISO 7/8). | Contamination control, environmental monitoring. |
| Process Control | Manual, variable parameters. | Fully defined, validated, and documented SOPs. | Defining critical process parameters (CPPs) and ensuring reproducibility. |
| Starting Materials | Research-grade, variable quality. | GMP-grade, qualified suppliers, Certificate of Analysis (CoA). | Sourcing and qualifying GMP materials; cost increase. |
| Radiolabeling | Low activity (µCi-mCi), manual. | High activity (Ci), automated modules (e.g., Synthera, Trasis AllinOne). | Radiation safety, engineering controls, automated synthesis validation. |
| Quality Control (QC) | Limited, for characterization only. | Full, stringent, batch-release testing. | Implementing rapid, compendial (USP/Ph. Eur.) methods within short half-life constraints. |
| Documentation | Lab notebook. | Batch Manufacturing Record, Device Master File, QC reports. | Rigorous, real-time documentation adhering to ALCOA+ principles. |
| Regulatory Framework | IACUC, IRB for animal studies. | cGMP (21 CFR 212), Radioactive Drug Research Committee (RDRC) or IND application. | Preparing Chemistry, Manufacturing, and Controls (CMC) section for regulatory submission. |
GMP Production Protocol Framework: A Case Study for a [99mTc]- or [68Ga]-Labeled Tracer
This protocol outlines the core steps for producing a GMP-grade, bacteria-specific imaging agent, assuming a cold kit for reconstitution with a radioactive isotope (e.g., (^{99m})Tc) or a direct radiolabeling (e.g., with (^{68})Ga).
Protocol Title: GMP-Compliant Production of a Single Batch of [68Ga]Ga-DOTA-(Bacteria-Specific Ligand) for Human Use
Objective: To reproducibly manufacture a sterile, apyrogenic, and radiochemically pure dose of a gallium-68 labeled tracer under cGMP.
I. Pre-Production Activities
- Personnel & Environment: All personnel must be GMP-trained. Perform area clearance and confirm cleanroom (ISO 8) and laminar flow hood (ISO 5) certifications are current. Record temperature and pressure differentials.
- Materials Release: Verify CoAs for all starting materials (GMP-grade ligand, buffer salts, sterilizing filters). Confirm the (^{68})Ge/(^{68})Ga generator eluent QC (radionuclidic purity, (^{68})Ge breakthrough < 0.001%).
- Equipment Preparation: Perform pre-production checks on the automated synthesis module (e.g., Scintomics GRP or IASON GGP). Assemble sterile, single-use fluid paths and reagent kits. Perform a pressure-hold test.
II. Automated Synthesis Procedure
- Elution: Elute the (^{68})Ge/(^{68})Ga generator with 0.1 M HCl directly into the synthesis module.
- Purification: Pass the eluate through a cation-exchange cartridge (e.g., Strata-XC) to pre-concentrate and purify (^{68})Ga(^{3+}).
- Labeling: Elute (^{68})Ga(^{3+}) with a mixture of 5 M NaCl/0.1 M HCl directly into the reaction vial containing the GMP-grade precursor ligand (e.g., 30 µg DOTA-TMP in sodium acetate buffer, pH 4.5). Heat at 95°C for 5-10 minutes.
- Formulation: Cool the reaction mixture. Pass through a C18 cartridge to trap the labeled product. Wash with sterile water. Elute the final product with ethanol/water or saline into a sterile, evacuated vial containing a phosphate buffer for final pH adjustment (~7.4).
- Sterile Filtration: Terminally filter the final product through a 0.22 µm sterile filter into a final, labeled, sterile dose vial.
III. In-Process and Release Quality Control Tests All tests must be performed per validated methods with pre-defined acceptance criteria. Results must be documented in the Batch Record before release.
Table 2: Essential QC Tests for a (^{68})Ga-Labeled Tracer Batch Release
| Test Parameter | Method | Acceptance Criteria | Typical Result | Rationale |
|---|---|---|---|---|
| Appearance | Visual inspection | Clear, colorless, particulate-free. | Complies. | Sterility and formulation integrity. |
| pH | pH strip or micro-electrode | 4.5 - 8.5. | ~7.2. | Physiological compatibility. |
| Radionuclidic Identity | Half-life measurement (γ-detector) | 62.0 - 69.1 min (T1/2 of (^{68})Ga). | ~68 min. | Confirms correct isotope. |
| Radiochemical Purity (RCP) | Radio-iTLC or HPLC | ≥ 95% (product-specific). | ≥ 98%. | Measures fraction of radioactivity bound to intended ligand. |
| Chemical Purity | HPLC (UV/Vis) | Ligand-related impurities < 5 µg/dose. | < 2 µg/dose. | Ensures low chemical impurity burden. |
| Radioisotopic Purity | γ-spectrometry | (^{68})Ge breakthrough ≤ 0.001%. | ≤ 0.0005%. | Limits long-lived impurity dose. |
| Sterility | Ph. Eur. 2.6.1 (Bactec) | Sterile. | Tested, read post-release. | Mandatory safety test (14-day incubation). |
| Bacterial Endotoxins | LAL test | ≤ 175 EU/V (V = max dose volume). | < 5 EU/mL. | Ensures product is apyrogenic. |
| Filter Integrity | Bubble-point test (post-use) | > Manufacturer's spec. | > 50 psi. | Verifies sterility assurance. |
IV. Post-Production
- Batch Release: The Qualified Person (QP) or authorized Pharmacist reviews the complete Batch Record and QC data. The product can be released for administration under an RDRC or IND before sterility test results are available, provided all other tests pass and aseptic conditions were validated.
- Dispensing: The final product vial is assayed in a dose calibrator, and individual patient doses are drawn aseptically into sterile syringes, labeled, and shielded.
- Documentation: All records (SOPs, BMR, QC reports, environmental monitoring) are archived for the regulatory retention period.
Visualizing the GMP Production Workflow and Regulatory Pathway
Diagram Title: GMP Production and Regulatory Pathway for Imaging Probes
Diagram Title: Single Batch GMP Production and QC Workflow
The Scientist's Toolkit: Essential Reagent Solutions & Materials
Table 3: Key Research Reagent Solutions for Probe Development & GMP Translation
| Item/Category | Function & Role in Development | GMP-Production Considerations |
|---|---|---|
| Precursor Ligand (e.g., DOTA-TMP, maltodextrin derivative) | The targeting molecule, chelated to the radiometal. Defines specificity and pharmacokinetics. | Must be sourced as a GMP-grade active pharmaceutical ingredient (API). Requires full characterization (NMR, MS, HPLC), stability data, and a Drug Master File (DMF). |
| Radionuclide (e.g., (^{68})Ge/(^{68})Ga generator, (^{99})Mo/(^{99m})Tc generator) | Source of radioactivity for imaging. | Generator must be qualified/approved for clinical use. Eluate must be tested for radionuclidic purity and breakthrough before use in GMP batch. |
| Buffer Systems (e.g., Sodium Acetate, HEPES, Phosphate) | Controls pH during radiolabeling and final formulation for physiological compatibility. | Must be sterile, GMP-grade, and pyrogen-free. Buffer composition is a critical process parameter. |
| Solid-Phase Extraction (SPE) Cartridges (e.g., C18, Cation Exchange) | Purifies the radionuclide (pre-concentration) and/or the final labeled product. | Must be sterile, single-use, and compendial (USP/Ph. Eur.). The elution profile must be validated. |
| Sterilizing Grade Filter (0.22 µm PES or PVDF membrane) | Ensures sterility of the final product vial. | Integrity must be tested pre- and post-production (bubble point test). Single-use, sterile. |
| Quality Control Kits (iTLC strips, HPLC columns, LAL reagents) | Used to perform release tests (RCP, chemical purity, endotoxins). | Methods must be validated for the specific product. Kits should be qualified. |
| Final Product Vial & Septa | Container-closure system for the sterile dose. | Must be sterile, pyrogen-free, and compatible with the formulation. Validated for leachables/extractables. |
Proof and Performance: Validating Probes and Comparing Clinical Utility
Within the pursuit of developing bacteria-specific molecular imaging probes for precise infection localization, new agents must be rigorously validated against established clinical standards. [18F]Fluorodeoxyglucose Positron Emission Tomography (FDG-PET) and Radiolabeled Leukocyte Scintigraphy (e.g., 99mTc-HMPAO-WBC SPECT/CT) represent the current imaging gold standards for infection and inflammation. This document outlines application notes and detailed protocols for using these modalities as benchmarks in preclinical and translational research.
Quantitative Comparison of Gold Standards
Table 1: Key Characteristics of Infection Imaging Gold Standards
| Parameter | FDG-PET/CT | Radiolabeled Leukocyte Scintigraphy (99mTc-HMPAO-WBC) |
|---|---|---|
| Primary Tracer | [18F]FDG | Autologous leukocytes labeled with 99mTc-HMPAO |
| Imaging Mechanism | Uptake via glucose transporters (GLUT) in activated inflammatory cells (neutrophils, macrophages) & some microbes. | Direct imaging of recruited effector immune cells to site of infection. |
| Key Advantage | High sensitivity, excellent spatial resolution, rapid imaging (<2 hrs post-injection), quantitative (SUV). | High specificity for infection over sterile inflammation, established clinical track record. |
| Key Limitation | Low specificity; uptake in tumors, post-surgical sites, sterile inflammation. | Labor-intensive ex vivo cell handling, radioactive burden to personnel, lower resolution (SPECT), delay to imaging (2-24 hrs). |
| Typical Administered Activity (Human) | 185-370 MBq | 185-740 MBq (depends on labeling efficiency) |
| Optimal Imaging Timepoint | 60 ± 10 min post-injection | 2-4 hrs (early) and 20-24 hrs (delayed) post-reinjection |
| Sensitivity (Reported Range) | 85% - 98% | 68% - 95% (higher for chronic infection) |
| Specificity (Reported Range) | 75% - 85% | 75% - 100% |
| Role in Probe Validation | Benchmark for sensitivity and biodistribution. Negative control (sterile inflammation) for specificity testing. | Benchmark for specificity and cell trafficking patterns. |
Experimental Protocols for Benchmarking
Protocol 3.1: Preclinical Murine Model of Bacterial Infection for Probe Comparison
- Objective: To compare uptake of a novel bacteria-specific probe against FDG and radiolabeled leukocytes in a controlled infection model.
- Materials:
- Animal model (e.g., C57BL/6 mouse).
- Bacterial strain (e.g., Staphylococcus aureus GFP-lux).
- [18F]FDG, 99mTc-HMPAO, novel probe (e.g., [68Ga]Ga-siderophore).
- MicroPET/CT or PET/SPECT/CT scanner.
- Gamma counter, biodistribution supplies.
- Procedure:
- Infection Induction: Anaesthetize mouse. Create a subcutaneous infection in the left flank (e.g., 1x10^7 CFU in 50 µL PBS + 10% Bacto gelatin). Inject sterile PBS+gelatin in right flank as sterile inflammation control.
- Group Allocation: Randomize animals into imaging groups (FDG, WBC, Novel Probe; n≥5/group).
- FDG Imaging (Day 1):
- Fast animals for 4-6 hrs.
- Inject ~3.7-7.4 MBq [18F]FDG via tail vein.
- Image at 60 min p.i. under anesthesia (10-min static PET, followed by CT).
- Radiolabeled Leukocyte Imaging (Day 2-3):
- Leukocyte Isolation: Draw blood (~0.8-1 mL) from a donor mouse via cardiac puncture into heparin. Isolate leukocyte-rich buffy coat via density gradient centrifugation (e.g., Histopaque).
- Cell Labeling: Incubate cells with 37-74 MBq 99mTc-HMPAO for 15 min. Stop reaction, wash cells x3 to remove free tracer.
- Reinjection & Imaging: Inject labeled cells (~5-10x10^6 cells) into the infected recipient mouse via tail vein. Acquire SPECT/CT scans at 4 h and 20 h p.i.
- Novel Probe Imaging (Day 4-5): Inject formulated novel probe (equivalent activity). Image at its optimal timepoint (e.g., 2 h p.i.).
- Ex Vivo Analysis: Euthanize animals immediately after final scan. Harvest organs (infection site, control site, blood, muscle, liver, spleen, kidney, bone). Weigh and measure radioactivity in gamma counter. Calculate % injected dose per gram (%ID/g).
- Data Analysis: Calculate target-to-background ratios (TBR). Compare TBR and %ID/g of novel probe to gold standards using statistical tests (e.g., one-way ANOVA).
Protocol 3.2: In Vitro Specificity Assay (Human Cells)
- Objective: To assess tracer uptake in human neutrophils vs. bacterial cultures.
- Procedure:
- Isolate human neutrophils from healthy donor blood using density gradient.
- Prepare aliquots of target bacteria (e.g., S. aureus, E. coli) and control mammalian cells (neutrophils, macrophage cell line).
- Incubate cells/bacteria with respective tracer (FDG, novel probe) in appropriate buffer for 30-60 min at 37°C.
- Wash thoroughly to remove unbound tracer.
- Measure cell-associated radioactivity via gamma counting (for radiolabeled probes) or lysate scintillation counting (for 3H/14C-labeled probes).
- Normalize counts to protein content (µg). Express as % uptake relative to input.
Visualization: Experimental Workflow and Mechanism
Diagram 1: Benchmarking Workflow for Novel Probes
Diagram 2: Mechanisms of Tracer Accumulation at Infection Site
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Benchmarking Experiments
| Item | Function/Application in Benchmarking |
|---|---|
| [18F]FDG | The PET gold standard. Used to define maximum sensitivity for detecting infection/inflammation and for biodistribution comparison. |
| 99mTc-HMPAO (Exametazime) | Chelator for radiolabeling isolated leukocytes ex vivo. Enables SPECT imaging of neutrophil trafficking. |
| Histopaque-1077/1119 | Density gradient medium for isolating peripheral blood mononuclear cells (PBMCs) and neutrophils from human or murine blood. |
| Bacterial Strains (e.g., S. aureus Xen29, GFP-lux) | Bioluminescent strains enable parallel optical imaging to confirm infection viability and location, correlating with nuclear imaging. |
| Matrigel or Bacto Gelatin | Used to create a localized, persistent infection or sterile inflammation focus upon co-injection with bacteria or PBS. |
| Heparinized Blood Collection Tubes | Essential for collecting blood for autologous leukocyte isolation and labeling without clotting. |
| Gamma Counter with 511 keV & 140 keV Windows | For ex vivo biodistribution analysis of tissues following PET ([18F], 511 keV) and SPECT (99mTc, 140 keV) tracer administration. |
| Small Animal Image Analysis Software (e.g., PMOD, VivoQuant) | Enables co-registration of PET/SPECT/CT data, volumetric region-of-interest (ROI) analysis, and quantitative SUV/SUVmax calculation. |
Introduction Within the pursuit of bacteria-specific molecular imaging probes for infection localization, two dominant probe classes have emerged: antibiotic-based and metabolism-based agents. This application note provides a comparative analysis of these strategies, focusing on their mechanisms, performance metrics, and experimental protocols to guide research and development.
1. Comparative Performance Data The following tables summarize key quantitative characteristics of representative probes from each class, based on recent in vitro and preclinical in vivo studies.
Table 1: In Vitro & Physicochemical Properties
| Property | [99mTc]Ciprofloxacin (Antibiotic) | [18F]FDG (Metabolic - Bacterial Uptake) | [11C]Para-aminobenzoic acid (PABA) (Metabolic) |
|---|---|---|---|
| Target | Bacterial DNA Gyrase/Topoisomerase IV | Hexokinase/ Glycolytic Pathway | Dihydropteroate Synthase (folic acid synthesis) |
| Bacterial Specificity | Moderate (can bind eukaryotic enzymes) | Low (high mammalian cell background) | High |
| Uptake Kinetics (tmax) | 60-120 minutes | < 30 minutes | 20-40 minutes |
| Key Interferent | Pre-existing antibiotic therapy | Host immune cells (neutrophils, macrophages) | Endogenous mammalian folate metabolism |
Table 2: Preclinical In Vivo Performance (Murine Infection Model)
| Metric | Antibiotic-Based Probe | Metabolic-Based Probe |
|---|---|---|
| Target-to-Background Ratio | 1.5 - 3.0 | 2.0 - 8.0+ |
| Sensitivity | High | Moderate to High |
| Specificity | Moderate | High (for specific probes) |
| Optimal Imaging Timepoint | 2-4 hours post-injection | 30-90 minutes p.i. |
| Ability to Discern Viability | Yes (targets live bacteria) | Yes (tracks active metabolism) |
2. Detailed Experimental Protocols
Protocol 2.1: In Vitro Specificity & Binding Assay for Antibiotic Probes Objective: To evaluate the specific binding of a radiolabeled antibiotic probe to live vs. heat-killed bacteria. Reagents: Radiolabeled antibiotic probe (e.g., [³H] or fluorescent-tagged vancomycin), target bacterial culture (e.g., S. aureus), control mammalian cells (e.g., RAW 264.7 macrophages), PBS, scintillation cocktail/plate reader. Procedure:
- Culture bacteria to mid-log phase. Prepare aliquots: live bacteria and heat-killed (80°C, 20 min).
- Wash bacterial pellets and mammalian cells 3x with PBS. Adjust to 1x10⁸ CFU/mL (bacteria) or 1x10⁶ cells/mL.
- In a 96-well plate, incubate 100 µL of each suspension with 100 µL of probe at varying concentrations (0.1-10 µM) for 30-60 min at 37°C.
- Centrifuge plates (3000 x g, 5 min). Wash pellets 3x with PBS.
- For radioactive probes: transfer pellets to vials, add scintillation fluid, count. For fluorescent probes: resuspend in PBS, measure fluorescence.
- Calculate specific binding by subtracting non-specific binding (measured in heat-killed samples) from total binding.
Protocol 2.2: Metabolic Probe Uptake Kinetics Assay Objective: To quantify the time-dependent uptake of a radiolabeled metabolic precursor (e.g., [11C]PABA) in bacteria. Reagents: [11C]PABA (or analogous tracer), bacterial cultures, stop buffer (ice-cold PBS with 0.1% unlabeled substrate), filtration unit (0.22 µm filters). Procedure:
- Grow bacteria to desired growth phase (log vs. stationary).
- At t=0, add tracer (≈37 kBq/µmol) to bacterial suspension. Maintain under optimal growth conditions.
- At timepoints (1, 5, 15, 30, 60 min), withdraw 1 mL aliquots and immediately vacuum-filter through 0.22 µm membranes.
- Wash filter 3x rapidly with 5 mL ice-cold stop buffer.
- Place filter in gamma counter or measure radioactivity with appropriate detector.
- Plot uptake (pmol/mg protein) vs. time to derive kinetic constants.
3. Visualizing Core Pathways & Workflows
Title: Antibiotic Probe Mechanism of Action
Title: Metabolic Probe Uptake and Trapping Pathway
Title: Head-to-Head Probe Evaluation Workflow
4. The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in Probe Evaluation |
|---|---|
| Radiolabeled Precursors | (e.g., [11C]CO2, [18F]F-, [99mTc]O4-) Used for synthesizing imaging probes with PET/SPECT isotopes. |
| Bacterial Strain Panels | Include Gram-positive (e.g., S. aureus), Gram-negative (e.g., E. coli), and resistant strains for specificity screening. |
| Heat-Killed Bacteria Preparations | Critical negative control to differentiate specific binding/uptake from non-specific probe accumulation. |
| Mammalian Cell Lines | (e.g., macrophages, fibroblasts) Assess off-target probe uptake and eukaryotic toxicity. |
| Animal Infection Models | Typically murine (thigh, lung) with established bacterial burdens for in vivo validation. |
| Gamma Counter / PET Scanner | Essential for quantifying radiotracer uptake in ex vivo samples and in vivo imaging. |
| Size-Exclusion Microfilters | (0.22 µm) For rapid separation of bacteria from incubation medium in uptake/binding assays. |
| Specific Metabolic Inhibitors | (e.g., competition with unlabeled substrate) To confirm enzymatic specificity of metabolic probe uptake. |
Within the thesis research on bacteria-specific molecular imaging probes for infection localization, rigorous validation is paramount. This document outlines the critical validation metrics—sensitivity, specificity, and biodistribution—and provides standardized protocols for their quantification. These metrics are essential for differentiating target bacterial infections from sterile inflammation and non-target tissues, directly impacting the probe's diagnostic accuracy and translational potential.
Core Validation Metrics: Definitions and Calculations
Sensitivity (True Positive Rate): The probability that the imaging test correctly identifies an animal/model with a confirmed bacterial infection. It measures the probe's ability to detect the target when it is present. Formula: Sensitivity = (True Positives) / (True Positives + False Negatives)
Specificity (True Negative Rate): The probability that the imaging test correctly identifies an animal/model without a bacterial infection (e.g., sterile inflammation or healthy tissue). It measures the probe's ability to avoid false positives. Formula: Specificity = (True Negatives) / (True Negatives + False Positives)
Biodistribution: The quantitative measurement of probe concentration over time in various organs and tissues, expressed as percentage of injected dose per gram of tissue (%ID/g). It defines pharmacokinetics, target engagement, and clearance pathways.
Table 1: Validation Metrics Summary Table
| Metric | Definition | Ideal Value for Probes | Key Influencing Factor |
|---|---|---|---|
| Analytical Sensitivity | Lowest detectable probe concentration in vitro. | Low nM to pM range | Probe affinity (Kd) for target. |
| Diagnostic Sensitivity | Ability to detect infection in vivo. | >90% | Target density, probe permeability, background clearance. |
| Diagnostic Specificity | Ability to distinguish infection from sterile inflammation. | >85% | Probe binding specificity, off-target uptake. |
| Target-to-Background Ratio (TBR) | Signal in target infection vs. non-target tissue. | >3 (Optical/SPECT); >1.5 (PET) | Biodistribution and clearance kinetics. |
Experimental Protocols
Protocol 3.1:In VitroDetermination of Binding Specificity and Affinity
Objective: Quantify probe affinity (Kd) for bacterial cells versus mammalian cells. Materials:
- Purified imaging probe (fluorescent or radiolabeled).
- Target bacterial strain (e.g., S. aureus).
- Control mammalian cell line (e.g., RAW 264.7 macrophages).
- Non-target bacterial strain.
- Binding buffer (e.g., PBS with 1% BSA).
- Flow cytometer or gamma/fluorescence plate reader.
Procedure:
- Prepare serial dilutions of the probe (e.g., 0.1 nM to 1000 nM).
- Incubate a fixed number of bacterial cells and mammalian cells with each probe concentration for 60 min at 37°C.
- Wash cells 3x with ice-cold buffer to remove unbound probe.
- Quantify cell-associated signal (fluorescence counts per cell or radioactivity in gamma counter).
- Perform saturation binding analysis using non-linear regression (e.g., one-site specific binding model in GraphPad Prism) to calculate Kd and Bmax.
Protocol 3.2:In VivoSensitivity/Specificity in a Murine Infection Model
Objective: Determine diagnostic sensitivity and specificity of the probe in a living subject. Materials:
- Mouse model: one group with bacterial infection (e.g., thigh muscle infection with P. aeruginosa), one with sterile inflammation (e.g., LPS or carrageenan injection), and healthy controls.
- Radiolabeled or fluorescent imaging probe.
- In vivo imaging system (e.g., PET/CT, SPECT/CT, or fluorescence imager).
- Microbiological plating for CFU determination (ground truth).
Procedure:
- Administer probe via tail vein injection at optimized time point post-infection/inflammation.
- Conduct longitudinal imaging at pre-defined time points (e.g., 1, 4, 24 h post-injection).
- Euthanize animals post-final scan. Excise target tissue (infection/inflammation site), contralateral healthy tissue, and major organs.
- Quantify ex vivo signal in tissues using a gamma counter or fluorescence spectrophotometer. Normalize to tissue weight for %ID/g.
- Confirm bacterial burden in infection group by homogenizing tissue and plating for CFU count.
- Calculate Metrics: Define a positive imaging result using a threshold TBR > 2. Compare imaging results to CFU-positive (True Infection) and sterile inflammation/healthy (True Negative) status to populate a 2x2 contingency table and calculate sensitivity and specificity.
Protocol 3.3: Comprehensive Biodistribution and Pharmacokinetics
Objective: Quantify the temporal distribution and clearance of the probe. Materials: As per Protocol 3.2.
Procedure:
- At multiple time points post-probe injection (e.g., 5 min, 30 min, 2 h, 24 h), euthanize cohorts of animals (n=5 per time point).
- Collect blood, major organs (heart, lungs, liver, spleen, kidneys), muscle, bone, and infection/inflammation site.
- Weigh all tissues and measure radioactivity or fluorescence intensity.
- Calculate %ID/g for each tissue and time point.
- Plot biodistribution curves and calculate pharmacokinetic parameters (e.g., elimination half-life from blood, peak uptake time in target).
Table 2: Exemplar Biodistribution Data for a 99mTc-Labeled Antibiotic Probe at 4h Post-Injection
| Tissue | %ID/g (Mean ± SD) | Infection Model (TBR) | Sterile Inflammation Model (TBR) |
|---|---|---|---|
| Infected Thigh Muscle | 5.67 ± 0.89 | (Reference = 1.0) | N/A |
| Inflamed Thigh Muscle | 1.22 ± 0.31 | N/A | (Reference = 1.0) |
| Contralateral Muscle | 0.45 ± 0.12 | 12.6 | 2.7 |
| Liver | 15.23 ± 2.45 | N/A | N/A |
| Kidneys | 8.91 ± 1.67 | N/A | N/A |
| Blood | 0.89 ± 0.23 | N/A | N/A |
Visualizations
In Vivo Probe Validation Workflow
Factors Influencing Sensitivity and Specificity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Probe Validation
| Item | Function & Application | Example/Supplier |
|---|---|---|
| Bacteria-Specific Probe | Core agent binding to microbial targets (enzymes, cell wall). | Custom-synthesized vancomycin-800CW or [18F]FDG. |
| Animal Disease Models | Provide physiologically relevant infection and inflammation controls. | Murine thigh infection (bacterial) vs. carrageenan-induced sterile inflammation. |
| Multimodal Imaging Systems | Non-invasive, longitudinal quantification of probe biodistribution and accumulation. | PET/CT, SPECT/CT, Fluorescence Molecular Tomography (FMT). |
| Gamma Counter / Plate Reader | Ex vivo quantitative measurement of radiolabeled or fluorescent probe in tissues. | PerkinElmer Wizard2 γ-counter, BioTek Synergy plate reader. |
| Saturation Binding Analysis Software | Calculate binding affinity (Kd) and receptor density (Bmax) from in vitro data. | GraphPad Prism (One site -- Specific binding model). |
| CFU Enumeration Materials | Establish "ground truth" bacterial burden for sensitivity calculation. | Tryptic Soy Agar plates, tissue homogenizer. |
This document provides detailed application notes and experimental protocols for bacteria-specific molecular imaging of three critical infections: orthopedic implant-associated infections, infective endocarditis, and bacterial pneumonia. This work is framed within a broader thesis on developing and applying pathogen-specific probes for precise infection localization, aiming to improve diagnostic accuracy and therapeutic monitoring in preclinical research.
Orthopedic Implant-Associated Infection (Staphylococcus aureus)
Application Notes
Imaging of orthopedic infections, particularly those involving biofilm-forming Staphylococcus aureus on implants, requires probes that can penetrate the extracellular polymeric substance and target conserved bacterial surface components. Current research focuses on overcoming the challenge of differentiating microbial colonization from sterile inflammation.
Table 1: Performance Metrics of Selected Probes for S. aureus Osteomyelitis Imaging
| Probe Name / Type | Target | Model (Mouse/Rat) | Imaging Modality | Target-to-Background Ratio (Mean ± SD) | Key Limitation |
|---|---|---|---|---|---|
| [68Ga]Ga-DOTA-Ubiquicidin (29-41) | Bacterial membrane (negative charge) | Murine thigh infection | PET/CT | 3.8 ± 0.5 | Uptake in sterile inflammation |
| 6″-[18F]Fluoromaltotriose | Bacterial maltodextrin transporter | Implant-associated osteomyelitis | PET/CT | 4.2 ± 0.7 | Specific to Enterobacteriaceae |
| Vancomycin-[89Zr]Zr-DFO | Peptidoglycan (D-Ala-D-Ala) | Tibial implant infection | PET/CT | 5.1 ± 1.2 | Slow blood clearance |
| PNA-FITC (anti-S. aureus) | S. aureus-specific rRNA | Femur osteomyelitis | Fluorescence (IVIS) | 2.9 ± 0.4 | Limited tissue penetration |
Detailed Experimental Protocol: [68Ga]Ga-DOTA-Ubiquicidin PET/CT Imaging of Implant Infection
Objective: To non-invasively localize a S. aureus orthopedic implant infection using a radiolabeled antimicrobial peptide fragment.
Materials:
- Animal Model: 8-10 week old C57BL/6J mouse.
- Infection: S. aureus (e.g., strain USA300 LAC; 1x10^5 CFU in 5 µL PBS) inoculated onto a sterile titanium pin surgically placed in the femur.
- Control: Sterile inflammation induced by implanting a pin coated with bovine serum albumin.
- Imaging Probe: [68Ga]Ga-DOTA-Ubiquicidin(29-41) (synthesized per GMP-like guidelines, ~10-15 MBq per mouse).
- Imaging System: Micro-PET/CT scanner.
Procedure:
- Model Establishment (Day -7): Perform aseptic orthopedic surgery to implant a titanium pin into the intramedullary canal of the left femur. Inoculate with S. aureus (infection group) or PBS/BSA (sham group). Administer post-op analgesia.
- Probe Preparation (Day 0): Elute 68Ga from a 68Ge/68Ga generator. Label the DOTA-Ubiquicidin(29-41) precursor following a standardized protocol (pH 3.5-3.8, 95°C, 10 min). Purify via C18 solid-phase extraction. Confirm radiochemical purity (>95%) by radio-TLC.
- Probe Administration: Inject ~12 MBq of the purified probe via the tail vein.
- Image Acquisition: At 60 minutes post-injection (pi), anesthetize the mouse (2% isoflurane). Acquire a static 10-minute PET scan followed by a low-dose CT scan for anatomical co-registration. Maintain body temperature at 37°C.
- Image Analysis: Reconstruct PET data using an OSEM algorithm. Co-register with CT. Define volumes of interest (VOIs) over the infected/implanted femur and the contralateral uninfected femur. Calculate standardized uptake values (SUVmean and SUVmax). Express infection uptake as a ratio to contralateral muscle (Target-to-Background Ratio, TBR).
- Ex Vivo Validation: Euthanize mouse post-scan. Harvest and weigh the implanted and control femurs. Measure radioactivity in a gamma counter to determine percentage injected dose per gram (%ID/g). Homogenize bones for quantitative plating to determine CFU/g tissue. Perform correlative histology (Gram stain, H&E).
Infective Endocarditis (Streptococcus spp. and S. aureus)
Application Notes
Imaging infective endocarditis (IE) necessitates high-sensitivity probes due to the small, deep-seated vegetations on heart valves. The primary goal is to distinguish septic vegetations from sterile thrombi. Probes are evaluated based on their ability to detect microcolonies early and monitor antibiotic efficacy.
Table 2: Imaging Probes in Preclinical Models of Infective Endocarditis
| Probe Name / Type | Target Pathogen | Animal Model | Imaging Modality | Signal in Vegetation vs. Sterile Thrombus (Fold Change) | Best Application |
|---|---|---|---|---|---|
| [99mTc]Tc-Contuscept | Viable bacteria / fungi | Rat catheter-induced IE | SPECT/CT | 2.5-fold | Broad-spectrum detection |
| [18F]FDG | Inflammatory cell uptake | Rabbit IE model | PET/CT | High, non-specific | Monitoring therapy response |
| Fluorescent Vancomycin (VAN-FL) | Gram-positive peptidoglycan | Murine IE | Intravital microscopy | 3.1-fold | Real-time valve imaging |
| Anti-GPC3-[64Cu]Cu-NOTA | Streptococcus gallolyticus surface antigen | Rat IE | PET/MRI | 4.0-fold | Pathogen-specific imaging |
Detailed Experimental Protocol: Fluorescent Vancomycin (VAN-FL) Intravital Microscopy of Murine Endocarditis
Objective: To visualize the early adhesion and colonization of Streptococcus oralis on mechanically damaged murine aortic valves using a fluorescently tagged antibiotic.
Materials:
- Animal Model: 10-12 week old wild-type mouse.
- Infection: Streptococcus oralis (e.g., strain ATCC 35037; 1x10^7 CFU in 200 µL saline) administered intravenously.
- Valve Injury: Transverse aortic constriction surgery or direct valve wire-scratch 24h prior to infection to create sterile vegetation.
- Probe: Vancomycin conjugated to a near-infrared fluorophore (e.g., Vanco-IR800CW, 2 nmol per mouse).
- Imaging System: Intravital microscope with a thoracic window or epifluorescence imaging system for explanted hearts.
Procedure:
- Valve Damage (Day -1): Anesthetize mouse. Perform a minimally invasive surgery to induce sterile mechanical damage to the aortic valve (e.g., via a guidewire inserted through the carotid artery). Close the incision.
- Infection & Probe Administration (Day 0): Inject S. oralis suspension intravenously. At 30 minutes post-infection, inject VAN-FL probe intravenously.
- Intravital Imaging (Day 1 or 2): Re-anesthetize the mouse. For thoracic window imaging, perform a surgical procedure to exteriorize the heart and aortic arch. Mount the mouse under a confocal or multiphoton microscope. Acquire time-lapse or static images of the aortic valve area using appropriate laser/excitation for the fluorophore. Maintain physiological conditions.
- Ex Vivo Analysis: Euthanize the mouse. Excise the heart, rinse in PBS, and image en bloc using a fluorescence macro-imager (e.g., IVIS Spectrum) to quantify total vegetation fluorescence. Open the heart under a dissection microscope, carefully excise the vegetation, and process for CFU enumeration and fluorescence microscopy on tissue sections.
Bacterial Pneumonia (Pseudomonas aeruginosa & Klebsiella pneumoniae)
Application Notes
Imaging pulmonary infections involves challenges related to aerosol delivery of probes, rapid mucociliary clearance, and overlapping signals from alveolar inflammation. Probes targeting species-specific virulence factors or siderophore systems are under investigation.
Table 3: Probes for Imaging Gram-Negative Bacterial Pneumonia
| Probe Name / Type | Target / Mechanism | Pathogen | Imaging Modality | Key Finding (Infected vs. Control Lung) | Status |
|---|---|---|---|---|---|
| [68Ga]Ga-FSC | Bacterial iron transport (pyoverdine) | Pseudomonas aeruginosa | PET/CT | 15-fold higher %ID/g | Preclinical |
| MAL-[11C] | Maltodextrin metabolism | Klebsiella pneumoniae | PET/CT | Specific uptake abolished by cold maltose | Preclinical |
| Ciprofloxacin-[99mTc]Tc | DNA gyrase (bacterial viability) | Mixed G- pneumonia | SPECT | Sensitivity: 85%, Specificity: 72% | Clinical trial phase II |
| Anti-LPS mAb-[64Cu]Cu | Lipopolysaccharide core | K. pneumoniae | PET/MRI | Detects antibiotic resistance (exposed LPS) | Preclinical |
Detailed Experimental Protocol: [68Ga]Ga-FSC PET/CT Imaging of P. aeruginosa Pneumonia
Objective: To exploit the bacterial siderophore system for specific imaging of P. aeruginosa lung infection.
Materials:
- Animal Model: Immunocompetent mouse (e.g., Balb/c).
- Infection: Clinical isolate of P. aeruginosa (e.g., PAO1 or mucoid strain; 1x10^6 CFU in 40 µL PBS) administered via oropharyngeal aspiration under anesthesia.
- Control: Sterile PBS aspiration or K. pneumoniae infection.
- Probe: [68Ga]Ga-FSC (FSC = Fusarinine C-based chelator, a P. aeruginosa siderophore mimic).
- Imaging System: Micro-PET/CT.
Procedure:
- Induction of Pneumonia (Day -1): Anesthetize mouse with isoflurane. Suspend vertically and administer bacterial suspension or PBS via droplet instillation into the oropharynx, ensuring inhalation. Monitor until fully recovered.
- Probe Synthesis (Day 0): Radiolabel the FSC precursor with generator-eluted 68Ga in sodium acetate buffer (pH 4.5-5.0) at room temperature for 5-10 minutes. Pass through a 0.22 µm filter. Confirm purity.
- Imaging: At 24h post-infection, inject ~10 MBq of [68Ga]Ga-FSC via tail vein. At 90 minutes pi, acquire a 15-minute static PET scan followed by CT.
- Quantification: Segment the entire lungs using CT-based VOIs. Calculate lung SUVmean and total lung radioactivity (%ID). Compare to contralateral muscle or a reference tissue.
- Biodistribution & Validation: Euthanize mouse immediately after imaging. Harvest lungs, liver, spleen, kidneys, blood, and a muscle sample. Weigh and measure radioactivity in a gamma counter to determine %ID/g. Homogenize lungs for quantitative CFU plating. Perform correlative histopathology (e.g., H&E, Gram stain).
The Scientist's Toolkit: Key Research Reagent Solutions
| Item Name | Category | Function in Infection Imaging Research |
|---|---|---|
| DOTA-/NOTA-/DFO-based Chelators | Chemical Scaffold | Enable stable radiolabeling of targeting vectors (peptides, antibodies, siderophores) with diagnostic radionuclides (68Ga, 64Cu, 89Zr). |
| Near-Infrared Fluorophores (e.g., IRDye 800CW, Cy7) | Fluorescent Tag | Conjugate to antibodies, antibiotics, or PNAs for in vivo fluorescence imaging and intravital microscopy with deep tissue penetration. |
| Precise Bacterial Luciferase Operons (e.g., luxABCDE) | Reporter Gene | Engineer into bacteria for real-time, substrate-free bioluminescence imaging to track infection burden and location longitudinally. |
| Species-Specific Peptide Nucleic Acid (PNA) Probes | Nucleic Acid Mimic | Fluorescently labeled probes for fluorescence in situ hybridization (FISH) to identify specific pathogens in ex vivo tissue sections. |
| Conditionally Replicating Phages | Viral Vector | Deliver reporter genes (e.g., luciferase) specifically to replicating bacteria, amplifying signal for highly sensitive detection. |
| Humanized Mouse Models (e.g., NSG-SGM3) | Animal Model | Support engraftment of human immune cells to study infection pathophysiology and probe performance in a more human-relevant context. |
Diagrams
Title: Orthopedic Infection Imaging Workflow
Title: Core Principle of Bacteria-Specific Imaging
This Application Note, framed within a broader thesis on bacteria-specific molecular imaging probes for infection localization, details critical study design and endpoint considerations for navigating the regulatory pathway. The development of novel imaging probes, such as those targeting bacterial surface enzymes or metabolic pathways, requires meticulous planning to meet the standards of agencies like the U.S. FDA and EMA. Success hinges on robust preclinical validation and carefully constructed clinical trials that demonstrate safety, diagnostic accuracy, and clinical utility.
Key Regulatory Considerations and Quantitative Benchmarks
Based on current FDA (2023-2024) and EMA guidance for diagnostic radiopharmaceuticals and antimicrobials, the following benchmarks are critical for approval.
Table 1: Primary Regulatory Considerations for Bacteria-Specific Imaging Probes
| Regulatory Aspect | Key Requirement | Typical Benchmark / Threshold | Relevant Guidance Document |
|---|---|---|---|
| Preclinical Safety | Toxicology, Biodistribution, Dosimetry | No observed adverse effect level (NOAEL) established; Effective radiation dose < 30 mSv per administration | FDA: IND Content and Format; ICH S9 (R2) |
| Analytical Validation | Probe Purity, Stability, Radiochemical Purity | Radiochemical purity > 95%; Sterility, Apyrogenicity confirmed | USP <823>, <821>; Ph. Eur. 5.0 |
| Diagnostic Performance | Sensitivity & Specificity vs. Composite Reference Standard | Sensitivity > 80-85%; Specificity > 75-80% (Pivotal Trial) | FDA: Clinical Performance Assessment for Diagnostic Radiopharmaceuticals |
| Clinical Utility | Impact on Patient Management (e.g., Change in Care Plan) | Statistical demonstration of >20% change in management vs. standard of care | FDA: Determining Substantial Evidence of Effectiveness; EMA: Guideline on clinical evaluation of diagnostic agents |
| Risk-Benefit Assessment | Benefit (Accurate Localization) vs. Risk (Radiation, Misdiagnosis) | Net Benefit must outweigh risks; PPV/NPV analyses for clinical consequences | FDA Benefit-Risk Framework |
Table 2: Common Clinical Trial Endpoints for Infection Imaging Probes
| Endpoint Category | Primary Endpoint Example | Secondary/Exploratory Endpoint Examples |
|---|---|---|
| Efficacy (Diagnostic Accuracy) | Sensitivity & Specificity vs. Histopathology/Microbiology Composite Reference Standard (CRS) | Positive/Negative Predictive Value (PPV, NPV); Likelihood Ratios; Area Under the ROC Curve (AUC) |
| Efficacy (Clinical Impact) | Rate of Change in Patient Management Post-Scan (Therapeutic Decision) | Diagnostic Confidence (Reader Scale); Time to Correct Diagnosis; Reduction in Unnecessary Antibiotic Use or Interventions |
| Safety | Incidence of Adverse Events (AEs) and Serious Adverse Events (SAEs) related to the imaging probe | Abnormal Laboratory Findings; Vital Sign Changes; Radiation Dosimetry per organ |
Detailed Experimental Protocols
Protocol 1: Preclinical In Vivo Validation of Probe Specificity
Objective: To demonstrate specific binding and accumulation of the investigational imaging probe at sites of bacterial infection versus sterile inflammation. Materials: Animal model (e.g., murine thigh infection model), Isogenic bacterial strain (e.g., Staphylococcus aureus), Sterile inflammatory agent (e.g., λ-carrageenan), Radiolabeled bacteria-specific probe (e.g., [99mTc] or [68Ga]-labeled antibiotic/antibody mimic), Control imaging agent (e.g., [18F]FDG), Micro-PET/SPECT/CT scanner.
Procedure:
- Model Induction: In two cohorts of animals (n≥8/group), induce (a) a bacterial infection in the right thigh via intramuscular injection of 10^6 CFU of bacteria, and (b) a sterile inflammation in the left thigh via injection of carrageenan.
- Probe Administration: At peak inflammation (e.g., 24h post-induction), administer a standardized dose of the radiolabeled bacteria-specific probe via tail vein injection.
- Imaging: Acquire serial static or dynamic images at 1h, 2h, and 4h post-injection (p.i.) using the appropriate scanner (e.g., PET for Gallium-68).
- Ex Vivo Analysis: Following terminal imaging, euthanize animals. Harvest and weigh infected/inflamed tissue, contralateral control tissue, and key organs (liver, kidneys, spleen). Measure radioactivity in a gamma counter.
- Data Analysis: Calculate target-to-background ratios (TBR) and percent injected dose per gram of tissue (%ID/g). Statistically compare probe uptake in bacterial infection vs. sterile inflammation (using Student's t-test, p<0.05 significant). Perform autoradiography on tissue sections and co-register with histology (H&E, Gram stain).
Protocol 2: Clinical Pivotal Trial Design for Diagnostic Accuracy
Objective: To evaluate the diagnostic performance of the investigational probe in patients with suspected bacterial infection (e.g., diabetic foot osteomyelitis, prosthetic joint infection). Study Design: Prospective, multicenter, single-arm or comparative imaging trial.
Procedure:
- Patient Selection: Enroll adult patients with clinical suspicion of the target infection. Key exclusion criteria: pregnancy, uncontrolled comorbidities, recent use of high-dose antibiotics confounding reference standard.
- Investigational Scan: Administer the standardized activity of the bacteria-specific imaging probe (e.g., 185 MBq ± 10%). Acquire whole-body or targeted SPECT/PET images at the optimal time point determined from Phase I/II studies (e.g., 2-4h p.i.).
- Reference Standard: Within a pre-specified window (e.g., 7 days) of the scan, perform the Composite Reference Standard (CRS). The CRS is a pre-defined algorithm combining: a) Microbiological culture from image-guided biopsy or surgical tissue, b) Histopathological analysis, c) Clinical follow-up at 6 months for resolution with targeted therapy.
- Image Analysis: Three independent, blinded central readers, unaware of clinical and reference standard data, will assess the scans. They will categorize each suspected site as positive, negative, or equivocal for bacterial infection, and provide a confidence score (1-5 scale).
- Endpoint Assessment: Compare the imaging result (positive/negative) for each index lesion to the binary outcome (infection present/absent) determined by the CRS. Calculate per-patient and per-lesion sensitivity, specificity, PPV, NPV, and the area under the receiver operating characteristic (ROC) curve. Inter-reader agreement will be assessed using Fleiss' kappa.
Visualization Diagrams
Diagram Title: Roadmap from Probe Development to Regulatory Approval
Diagram Title: Pivotal Trial Diagnostic Accuracy Assessment Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Bacteria-Specific Imaging Probe Research
| Item | Function & Application | Example/Notes |
|---|---|---|
| Chelator-Linker Conjugates | Enables stable radiolabeling of targeting vectors (peptides, antibodies) with diagnostic radionuclides. | DOTA, NOTA, NODAGA, DFO for Ga-68, Zr-89, In-111, Cu-64. |
| Targeting Vector (Core) | Provides specificity for bacterial targets (enzymes, cell wall components). | Vancomycin-derivatives, UBI (Ubiquicidin) fragments, PNA (Peptide Nucleic Acids), bacteriophage proteins. |
| Control Probes (Non-specific) | Essential for validating specificity in preclinical models; control for increased vascular permeability/inflammation. | [18F]FDG (metabolic activity), Radiolabeled IgG (non-specific protein), scrambled peptide sequences. |
| Bacterial Strains & Animal Models | For in vitro binding assays and establishing in vivo infection models. | Reference strains (e.g., S. aureus ATCC 29213, P. aeruginosa PAO1); murine thigh, myositis, or implant infection models. |
| Sterile Inflammation Inducers | To create control models for differentiating infection from sterile inflammation. | λ-Carrageenan, Zymosan A, Lipopolysaccharide (LPS), damaged tissue (mechanical injury). |
| Radio-TLC/HPLC Systems | For quality control assessment of radiolabeled probes (radiochemical purity, stability). | ITLC-SG plates, Radio-HPLC with UV and gamma/radiodetectors. Required for every production run. |
| In Vivo Imaging System | For non-invasive, longitudinal assessment of probe biodistribution and target engagement. | Preclinical micro-PET/SPECT/CT or Optical Imaging (IVIS) systems. |
| Gamma Counter & Autoradiography | For precise ex vivo quantification of radioactivity and spatial localization in tissues. | PerkinElmer Wizard2; Phosphor Imager systems co-registered with histology slides. |
Conclusion
The development of bacteria-specific molecular imaging probes represents a paradigm shift towards precision diagnosis in infectious diseases. Foundational research has identified a robust set of bacterial targets, leading to diverse methodological strategies, from repurposed antibiotics to innovative metabolic probes. While optimization challenges around specificity, pharmacokinetics, and manufacturing persist, the field has demonstrated significant preclinical success and promising early clinical validation. When compared to traditional, inflammation-sensitive imaging, these probes offer the transformative potential to accurately localize infections, guide targeted interventions, and monitor treatment efficacy. Future directions must focus on advancing probes through clinical trials, expanding their spectrum to cover resistant and dormant bacteria, and integrating them with therapeutic ('theranostic') platforms. Success in this endeavor will not only improve patient outcomes but also strengthen antimicrobial stewardship in an era of rising resistance.